On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...

This week, Dr. Caroline Szczepanski sat down with the #ISCblog editors to share her exciting research on creating new materials that mimic the superadhesive or super-water resistant properties of natural surfaces. In her laboratory, Dr. Szczepanski studies and recreates surface properties that nature has perfected over thousands of years, and then harnesses and optimizes them for use in everyday products, such as contact lenses and rain boots. Read on to learn about how Dr. Szczepanski creates these surfaces in the laboratory.

Dr. Caroline Szczepanski
What is your field of study?
My degrees are all in chemical engineering, so I’m trained in the core principles associated with that discipline, including kinetics, thermodynamics, and process design. However, my research (and thus what I spend the majority of my time working on) is in materials science, which as a field focuses on designing and discovering new solid materials. In particular, I try to design and discover new materials made from polymers.
What influenced you to pursue this path of work/study? Broadly speaking, my parents were supportive of my pursuit of studies in science and engineering. My mother was a chemistry teacher – and the first Chemistry class I ever took (at the age of 15) was taught by her! She had worked as an industrial chemist prior to teaching, so I remember her telling me stories of mishaps and adventures in her laboratory, and I thought that sounded exciting. I definitely have her to thank for my interest in Chemistry.
What path (education, life experience, etc.) led you to where you are now?
One great thing about working in research and science in general is that things are constantly changing and evolving. Because of this, as you progress in your career, there are more and more factors that influence what you choose to study or devote your time to.
For me, I started doing research at my undergraduate institution (Lafayette College) because I needed a summer job! I found that I liked the work a lot, and the professor who was my mentor in that position encouraged me to apply to other research opportunities and eventually graduate school. I ended up at the University of Colorado for my PhD, and it was my work there that got me involved in polymer science and engineering, specifically studying polymer networks formed using light. After I defended my PhD, I was interested in doing more work in the field of polymer science, but wanted to expand the applications I was exploring and the techniques I was using in my research. I moved to Nice, France for a post-doctoral research position, where I studied the design of interfaces. During my post-doc I learned a lot about biomimicry and how it is used in numerous polymer applications. So now, in my position as an Assistant Research Professor in Chemical & Biological Engineering at Northwestern I combine different aspects of what I have learned at each of these different stages of my career into my current research.
What are polymers and why are polymer networks important? What are they used for? Why don’t monomers work as well in these cases?
Polymers are a class of macromolecules that are made up of many repeat units. Polymers are formed by combining thousands of repeat units (which exist as individual molecules called monomers) into a single molecule through a reaction that we call a polymerization.
For reference, the word polymer comes from Greek, with mer meaning “parts” and poly meaning “many”. A mono-mer, di-mer, tri-mer etc. indicate similar molecules made up from one, two, or three of these repeat units.
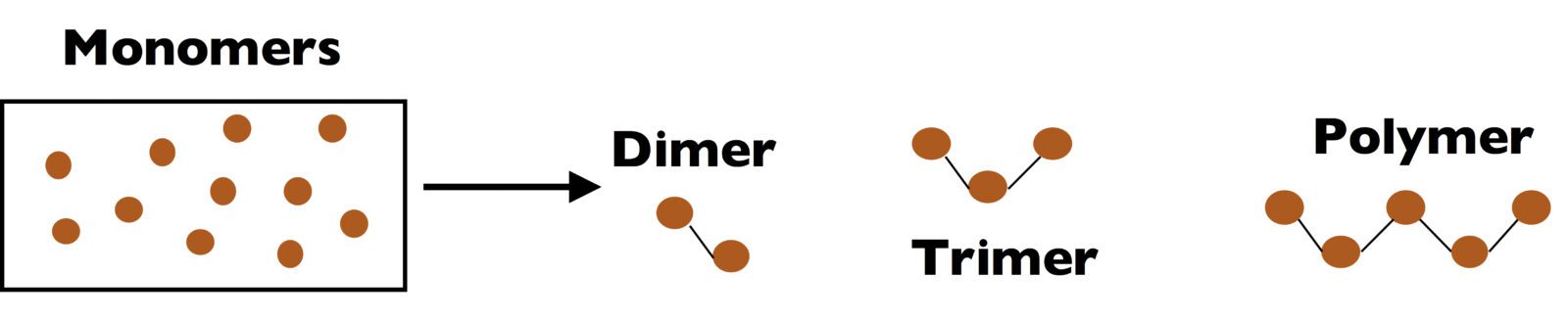
Polymers are special, because creating such a large macromolecule yields very unique properties and interesting materials. Polymers and polymer networks are used in everything from the bottles we drink from, the tires we drive on, the coatings on our cars. Even very small materials, like contact lenses, are made from polymers.
You mentioned on your website that you “unmix” materials with phase separation. Can you explain how that works and why it’s an essential part of your process?
When we design polymers and polymeric materials for a given application, there’s typically a list of attributes or characteristics that the final materials must have based on the desired use. This could mean you want your polymer to be very strong, but also water-repellant. Or maybe you want it be flexible but stable at really high temperatures. Either way, when there is such a profile it is typically insufficient to utilize a single monomer to build a polymer that addresses all of those different properties. For that reason, many polymer scientists and engineers look at ways to create heterogeneous polymeric materials. By having different polymers within one material, you can access different properties. Imagine if polymer A is flexible and polymer B has a high thermal stability – by combining the two in a single product you can achieve the desired profile.
Historically, the way engineers have combined different polymers is through mixing or blending. This process typically requires a lot of effort to do the mixing and the mixing is often done at high temperatures – meaning it requires a significant amount of energy to engineer the final material. I look at polymerization-induced phase separation, because it’s a process that is driven by slight incompatibilities between polymers that are made from different monomer units. Due to these incompatibilities, each component wants to segregate into a purer phase where it is compatible with other molecules exactly like itself. Using these incompatibilities to create heterogeneous materials requires less energy than physically mixing or blending two separate components. It is a more efficient method to combine multiple properties into a single material.
With phase separation, you are exploiting the same principle as when you see oil and vinegar separate. This separation is caused by the two different components wanting to be isolated so they can exist in a lower energy state. I design polymeric materials that undergo the same process, and form heterogeneous domains. By harnessing thermodynamics to control heterogeneous structure, materials can be designed much more efficiently.
Can you explain what biomimicry is? Why is mimicking biology important in creating polymers?
Biomimicry is the practice of using naturally occurring objects, materials, processes, forms, etc. to inspire current designs. In many ways, it’s a common practice that has been employed throughout our lives, but many of us are just simply unaware of the official name. The most obvious example is looking at an airplane, and seeing that it’s general form is similar to that of a bird. When imagining how it would be possible to build a vessel that could fly – we looked to nature for inspiration. In another case, the structure of Velcro was designed using biomimicry. The hook structure observed on cockle-burs are the inspiration for the structure of this type of adhesive. George de Mestral (the inventor) had observed that these plants would stay stuck onto different textiles, and mimicked that mechanism to create one of the most broadly used adhesives.
These are just a few examples amongst many, but the main advantage with biomimicry is that nature has been doing research for thousands of years. Evolution has found solutions for problems that different species have faced over time. If we look to nature, we might be able to find already established solutions to current engineering problems that are relevant in our modern world because nature tackled it a long time ago.
Mimicking biology is important in polymer science for a lot of reasons, but I’ll mention the one I’m most focused on. My area of research focuses on mimicking biological forms or structures on interfaces using polymers to create materials that could be used as coatings. Typically, the main purpose of a coating is to control the interaction at an interface between a material and an external environment. When designing coatings, it is often desirable for that surface to repel water, or for another application it might be necessary to capture a fluid or have the surface be strongly adhesive. Luckily, various plants and animals have surfaces – whether it is a leaf or a shell – which have these same properties. One well-known example is the lotus leaf, which repels water and is self-cleaning. With microscopy, we’ve observed that this leaf, as well as many other natural surfaces, actually have well-ordered micro and nanoscale features that lead to this property. For this reason, we now know that in order to create surfaces that have similar interactions as what we observe in nature, it is necessary to create well-patterned features on a micro or nano scale. Polymers, being easily processed and patterned are one way to create these types of surfaces. Patterns that have been explored with polymers include pillars, tubes, agglomerates, fibers, and bumps.
What are some examples of biological properties (ex: hydrophobicity) that you have incorporated into polymers?
My work has mostly focused on using biomimicry to control the wettability of surfaces and coatings made with polymers. Wettability refers to how a material interacts with a fluid, most often we think about water. Some of my past projects have focused on developing superhydrophobic surfaces, which are incredibly water repellent and self-cleaning (meaning, droplets roll right off a surface). Superhydrophobic surfaces are used as coatings for textiles (for example on rain boots or waterproof jackets), as well as on windshields of cars so they can repel water so you have decent visibility when driving through a rainstorm.
I’ve also worked on controlling the adhesion of different fluids to a surface. These types of surfaces can be used for water collection and filtration systems (such as those used in dry or isolated climates). As I mentioned, the lotus leaf has a self-cleaning property. However, the rose petal which also has a unique micro-structured surface, has very strong adhesion with water. This means that water droplets on a rose petal, despite looking like they have very little contact with the surface, have remarkably strong adhesion with the surface. I have worked to mimic rose-petal behavior in my research.

Rose Petal

Biomimicry of Rose Petals
Check out the rose petal mimic’s behavior in action!
Lastly, I have mimicked the strong adhesion observed on gecko toe-pads (which permits them to walk on vertical or inverted surfaces). This strong adhesion is due to nano-scale setae, which are pillar-like in shape. In my work I’ve been able to re-create strong adhesion by developing nano-tube features using polymers at an interface.

Nanotubes, Biomimicry of Gecko Toe Pads
How do you make these different interfaces from polymer networks?
I have used two different approaches to make polymeric interfaces. In some of my work, I have used a process called electropolymerization. With this process, I can create polymer films on a conducting surface (such as a metal like gold). The polymerization reaction occurs in a liquid solution of monomer and a solvent and is initiated by applying an electric potential to the liquid solution. This causes the monomers to oxidize in solution, making them reactive and causing them to combine with other monomers to create polymers. The polymers will eventually deposit onto the conducting surface as they become insoluble in this liquid solution, creating a coating on the electrode. With this process, the resulting interface can be manipulated by changing the solvent used as well as the electropolymerization parameters.
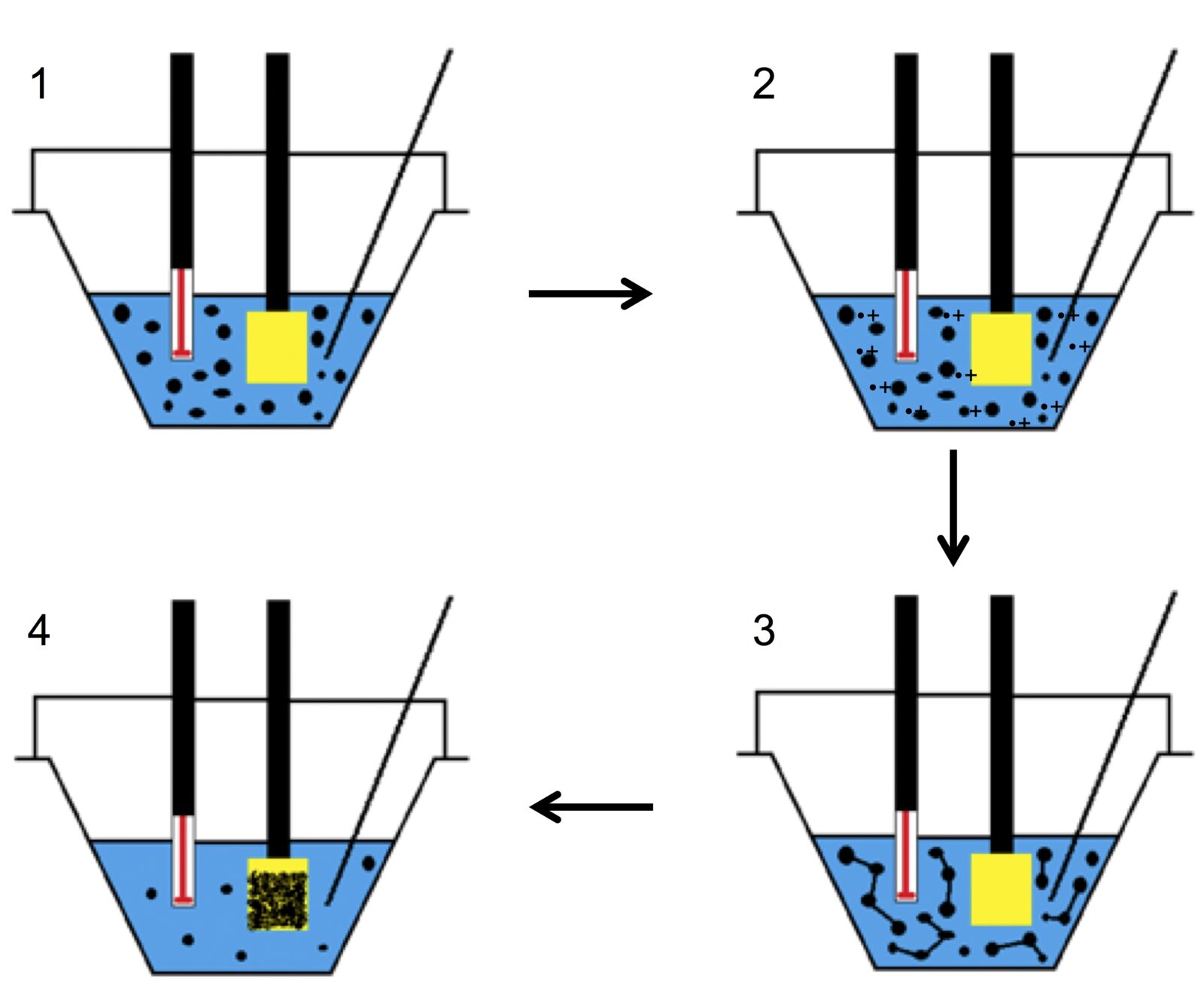
The Process of Electropolymerization
Another technique I use in a lot of my work is photo-initiated polymerization. Here, the polymerization reaction is started by exposing a solution of monomers to light. This technique is the same process that is used currently to create dental composites or “fillings” when you have a cavity. The energy from intense light can be used to excite small molecules called initiators which then kick-start a polymerization. I like to employ this technique because it’s very versatile. We can decide when and where the reaction happens (you can easily turn a light on or off, and easily decide where light will shine using different patterns). This is why this type of approach is becoming more and more attractive for applications varying from biomaterials (like the dental fillings) to industrial coatings and adhesives.
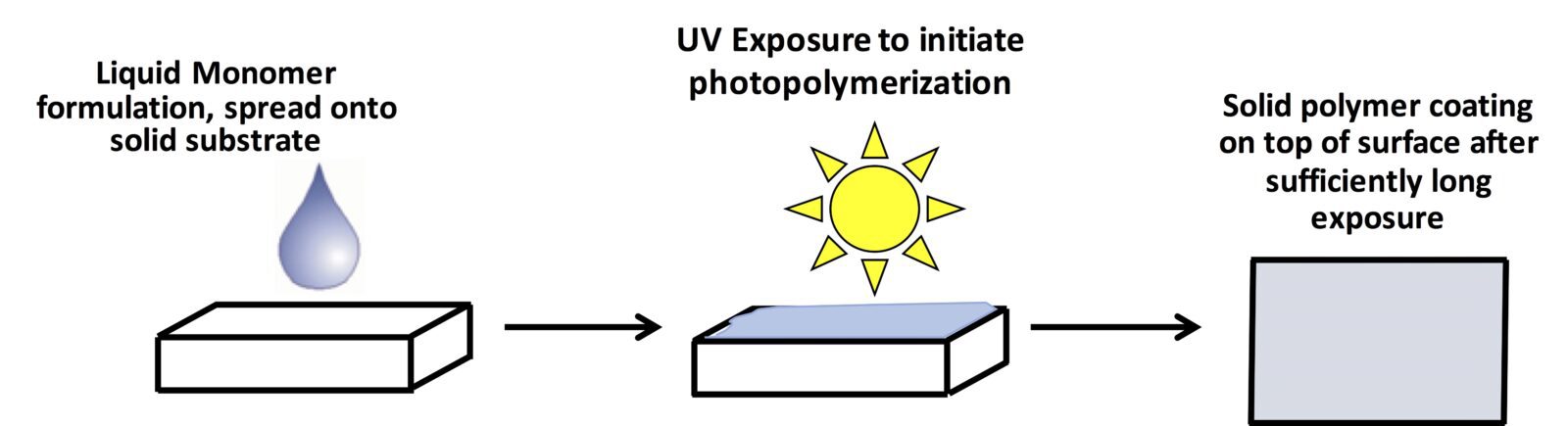
The Process of Photopolymerization
What is your earliest memory in a science class?
So this memory does not technically come from a classroom, but it’s along the same tone. As I mentioned my mother was a chemistry teacher. As a young kid I would always ask her “what she did in lab”. So, for Christmas when I was 7 or 8 she bought me a “starter chemistry kit” so we could do our own experiments at home! I remember the next day we did an experiment in our basement that involved doing a small reaction where the product was essentially table salt. We left our final product out to dry (and form the salt precipitates) overnight, and it was so amazing the next morning to see that what had been a liquid solution had turned into crystals! Needless to say I was hooked.
Can you give one piece of advice to aspiring scientists?
The first would be, don’t dismiss experiences that may seem a bit different than what you had envisioned for your future. It’s easy in academia or science to feel like you have a very direct plan and direction for your career path or project. While it’s good to have an outline and know where you want to go, sometimes unexpected experiences or side projects can lead to new ideas, partnerships, or problems that you would have never thought of before. I think too often people are so focused on a specific goal they get tunnel vision and forget about the bigger picture and often turn down opportunities (the chance to see an interesting seminar or go to a cool conference, or maybe visit a new lab) that may be more valuable than they realize.
Secondly, I would say don’t get discouraged by the failures. It is discouraging to have something go “unplanned” or against your original hypothesis, but that does not mean the result or the time you spent getting that result is invaluable. In fact it has a LOT of value. In science, failures are just as important as triumphs.
Interview of Caroline Szczepanski by Science Unsealed Editor, Dana Simmons.