On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...
Emily Collins has spent her life under the shadow of recurring bacterial infections. She spent a significant part of her childhood in the hospital, taking time off from her studies at her university, which cause her to lose her dream job of being a nurse. Emily suffers from chronic ESBL infections. ESBLs, or Extended-Spectrum-Beta-Lactamases, are bacterial chemicals that can break down a range of antibiotics, helping certain bacteria resist a large fraction of antibiotic treatments. As a result, these antibiotic-resistant infections are notoriously difficult to treat and can cause chronic, life-long infections.
Impact of Antibiotic-Resistant Bacteria
Antibiotic resistance is considered a global threat alongside climate change and war. According to the CDC, antibiotic resistant bacteria cause 23,000 deaths per year in the U.S. alone. Overuse and misuse of antibiotics, as well as a lower rate of new antibiotic development, has contributed to the rapid increase in antibiotic resistant bacteria. Therefore, scientists and doctors are now rushing to discover and implement new strategies to counter this rising threat.

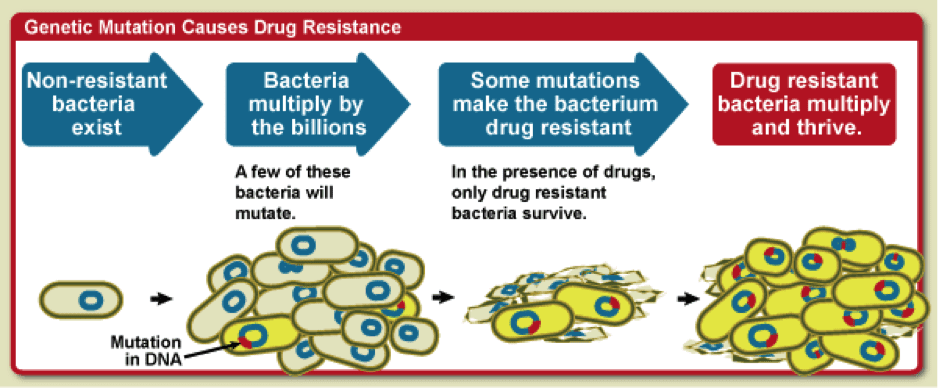
Figure 1: How antibiotic resistance can occur. Source.
Antibiotic resistance occurs in different ways: bacteria break down antibiotics to render them harmless, they pump antibiotics out of their cells before the drugs cause damage, or they change their outer structure so that the antibiotics cannot enter. “Bacteria have a big advantage on us as their numbers are much larger. Even if a very low percentage of them are resistant, they can survive antibiotic treatment,” explained Dr. Paul Hergenrother, Professor of Chemistry at the University of Illinois at Urbana-Champaign. Unfortunately, bacteria can pass on these defense mechanisms to other antibiotic sensitive bacteria. This information exchange, known as horizontal gene transfer, involves the movement of genetic material from one bacterium to another. Humans can only pass genetic information on to their offspring; horizontal gene transfer is as if you could give your genes for your hair color to your friend, so that their hair color would now match yours! Because bacteria can do horizontal gene transfer, they can transfer antibiotic resistance information to neighboring bacteria, thus creating an army of infections that are difficult to treat.
How do we prevent antibiotic-resistant bacteria?
Combating antibiotic resistance requires a multi-pronged approach. First, doctors need to reduce the number and type of antibiotics they prescribe. For example, antibiotics are ineffective against viral diseases such as sore throats and ear infections. Therefore, doctors need to confirm whether bacteria are causing a given infection. The antibiotics prescribed also depend on the type of bacteria. Antibiotic resistant bacteria require different classes of antibiotics, which ultimately saved Emily’s life. These classes are used as a last resort to avoid the possibility of antibiotic resistance. Therefore, they should not be used against antibiotic sensitive bacteria.
The second approach requires governments to incentivize the process of antibiotic development, which is less profitable than other classes of medicines. Discovering new antibiotics is an uphill task because scientists are still trying to understand the biological processes that occur in bacteria. The situation is complicated because bacteria can accumulate mutations, allowing them to evade the effect of antibiotics. Scientists are also focusing on improving the current antibiotics to make them more effective. For example, Dr. Hergenrother’s lab focuses on determining what chemical features enable antibiotics to enter bacteria.
The final approach involves strategies that target bacteria without the use of antibiotics. For example, hospitals are trying to reduce the rates of health-care acquired infections, which cause 99,000 deaths per year in the U.S. alone. When a patient as vulnerable as Emily is treated in a hospital, she can be exposed to pneumonia, urinary tract infections, or blood stream infections. Although the risk is considerably lowered by practices such as washing hands with soap water and alcohol, there is room for improvement. Companies like CuVerro are pushing for the use of copper surfaces in hospital equipment. Copper has historically been used to reduce bacterial numbers. Since most bacteria are unable to survive on copper surfaces, these can be used to reduce the transmission of diseases.
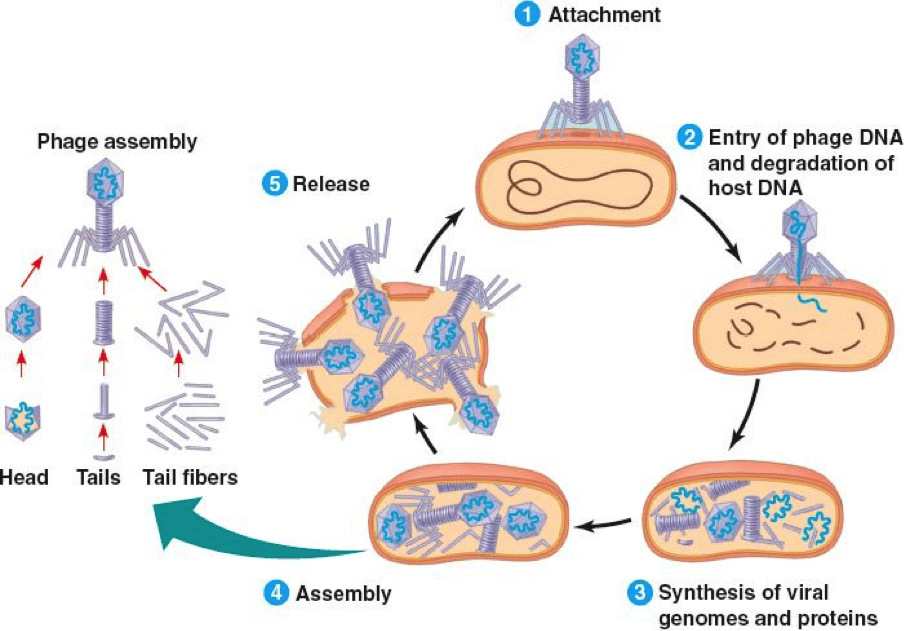
Figure 2: How bacteriophage infect and kill bacteria. Source.
Scientists are also trying to team up with an enemy of bacteria– bacteriophages. Bacteriophages are viruses that exclusively target and kill bacteria. They enter the bacteria and hijack the cells to form a virus-producing factory. Bacteria can evolve to prevent these processes, but bacteriophages also evolve to find a new way to invade the bacteria and kill them, offering a big advantage. Bacteriophage therapy was as early as 1910 but its clinical development declined after the discovery of antibiotics. Phage cocktails are now being developed, which target different cell processes in bacteria simultaneously. This treatment further reduces the likelihood of bacteria to evolve resistance. Unfortunately, phage therapy has not been approved by the FDA yet, but clinical trials are showing promising results with regard to the safety and efficacy of the treatment.
The direction forward with antibiotic-resistant bacteria
Bacterial resistance to treatments will always be a problem because of bacteria’s ability to mutate in order to survive. However, there are steps that can be taken to reduce the incidence of these mutations. Since we currently have a heavy dependence of antibiotics, which can cause havoc in patients like Emily, we need to introspect how we use antibiotics, how we can develop better antibiotics, and how we can try to move towards antibiotic-independent treatments.
Paul Hergenrother can be contacted at hergenro@illinois.edu.