On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...
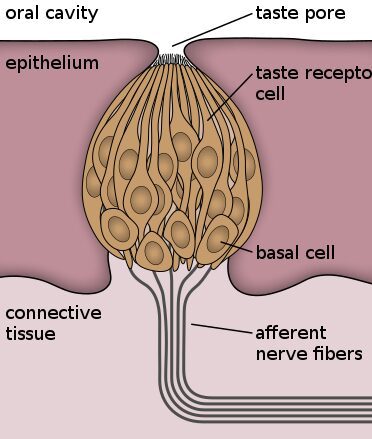
Have you ever wanted an instrument for determining which biological molecules are present in a mixture? Okay, unless you’re working in a lab, you probably haven’t had that desire. But the good news is, you have one anyway, and you take it with you wherever you go- it’s your tongue!
Every time we sit down to enjoy a meal, we’re conducting a small experiment in biochemical analysis.
Taste itself is comprised of many factors. Whether a meal is hot or cold, how spicy or ‘cool’ (think minty) it is, and the texture of the food will determine your overall enjoyment of the meal, largely based on personal preferences. However, underlying all these other components are the ‘basic’ tastes. These are the flavor categories you probably learned about in grade school- sweet, sour, salty, bitter, and savory.

Now, the ‘tongue map’ you also probably learned about in grade school is a myth. Every single taste bud (and you have somewhere between two and eight thousand) has taste receptors for each of these flavours. The map was made in 1901 and debunked by 1974, so you don’t have to crunch your chips on the side of your mouth. But there are unique components to each of the flavours we enjoy, and understanding what contributes to taste is a great illustration of how science affects our lives in ways we often don’t even consider!
Sweet Taste
Taste buds contain a wide variety of receptors called G-protein coupled receptors, or GPCRs. These receptors reside in the cell membrane, forming a communication line between the inside and the outside of the cell. The molecules in your meal that make it sweet fit into this receptor, changing its shape, which ultimately signals to your brain that you’ve eaten something “sweet.”
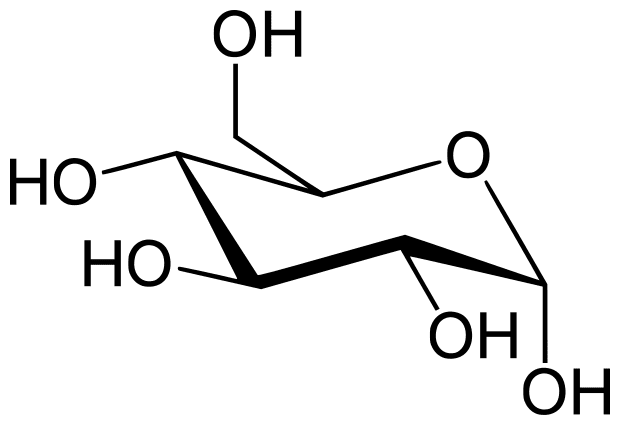
Sweet molecules are often sugars. On a molecular level, a sugar looks like this:
That’s glucose. It is a specific type of sugar that most organisms on earth have evolved to use as their main energy source. You can imagine why many people have a sweet tooth- until the modern abundance of mass-produced food, humans had to work hard to farm, hunt, or scavenge their food, and finding something sweet meant it had a lot of energy to keep them running on a full tank!
Sour Taste
Sourness is a result of the acidity of your food. Acidity comes from compounds that can donate a hydrogen ion (H+). This results in free H+ions, which can move from the outside to the inside of the cell without special receptors like sugars need. As the number of H+ions increases, the pH goes down, and sourness is increased. Water has a pH of 7 and is not sour at all (hopefully!) Lemons are incredibly sour because they have a lot of citric acid in them. Citric acid by itself makes a solution with a pH around 3. Since the pH scale (right) is logarithmic, that means lemons are 104 or 10,000 times more sour than water!
Salty
Saltiness of your food also comes from ions. These are ions are from larger atoms, which are too big to cross your cell membranes without help. Instead of using receptors, like sugars, they have special channels that allow them to enter the cell through passive diffusion. Table salt is sodium chloride, or NaCl, which dissolves easily into Na+ and Cl– ions. (Sodium is “Na” because it derives from the Latin word for salt, natrium.) It’s the sodium ions that tell our brain a food is salty.
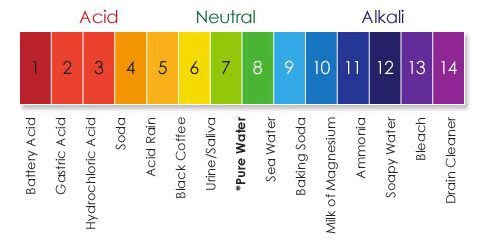
Sour Taste
Another ion that will taste salty to us is potassium, or K+. (Potassium also has a “neo-Latin” origin with its atomic symbol, from the word kallium.) Potassium is just below sodium on the periodic table, which means they have similar properties, most notably their single charge, but potassium is a bit bigger and less abundant. Sodium and potassium have an important relationship in your body.
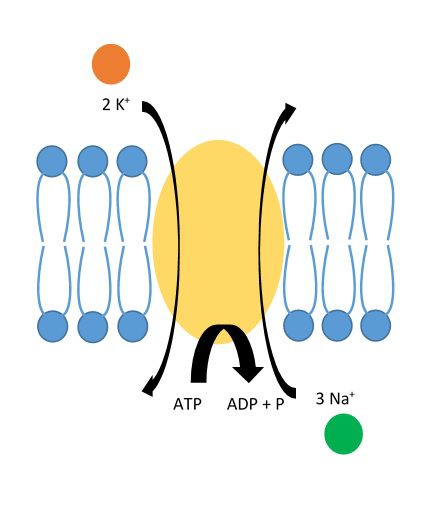
If left to the laws of diffusion, the amount of sodium and potassium on the inside vs. the outside of your cells would be equal. However, the amount of sodium in your body would mean that too much sodium is inside your cells. Conversely, the amount of potassium inside your cells would be too low. So your cells have special pumps to force these ions out/in against their concentration gradient. This pump moves three Na+ ions out at the same time as it moves two K+ ions in. It requires energy (in the form of ATP) to operate. This may seem like a simple principle, but understanding how it worked earned Jens Christian Skou a Nobel Prize in 1997!
Bitter
Our ability to discern things as bitter is of special interest to evolutionary biologists. They believe this taste is a result of natural selection, as bitter things tend to be hazardous to our health. A good example of this is strychnine, found in pesticides. Being sensitive to the bitter compounds in poisonous foods is quite an advantage when living the hunter/gatherer lifestyle!
There is a huge number of compounds that make food taste bitter, and thus we have a wide variety of receptors that determine bitter. Unlike sugars, bitter compounds can come in many shapes and sizes, so a single receptor is not sufficient to suss out every bitter flavour. Slight genetic differences in these receptors can affect how sensitive they are, and certain gene mutations lead to “super-tasters,” or people with a very sensitive palette. The specific receptors that control bitter taste are called “Taste receptors type 2” or TAS2Rs (scientists love acronyms that also sound like what they’re studying…). Studying gene variations in these types of receptors is also of interest to geneticists.
When bitter compounds activate their receptors, the cell’s Na+/K+pump is slowed down or turned off, which affects the overall balance of ions in your cells. This slight deregulation tells your brain you have eaten something bitter. This helps us understand why ‘bitter’ is often a precursor to poisoning- if you eat something that has the ability to turn off the cell’s ability to regulate itself, your cells will die- which doesn’t bode well for you!
Savory/Umami
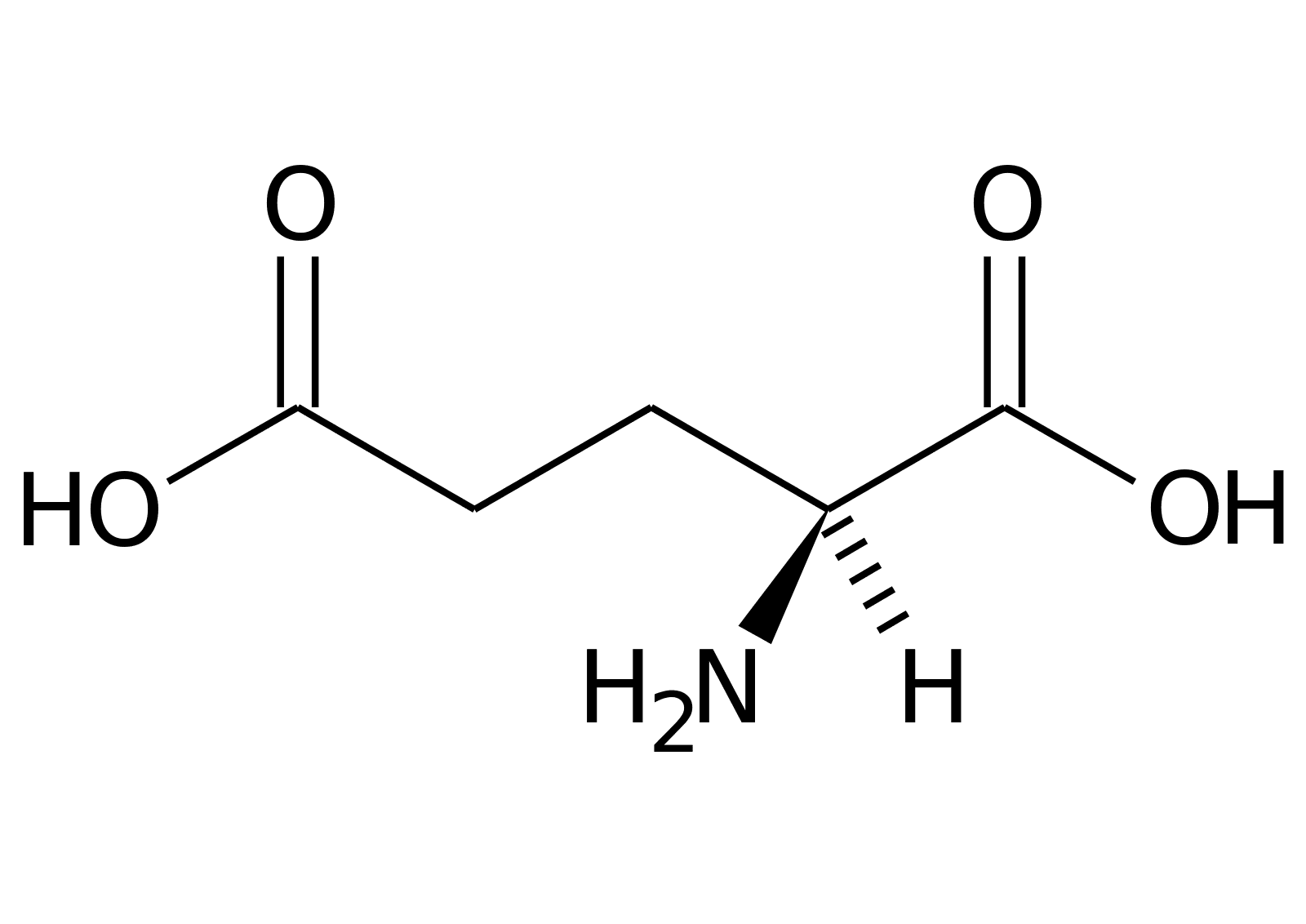
The Umami taste is considered a “recent” addition to the basic tastes, but the reality is that in Eastern cultures (such as Japan, where the name comes from), savoriness has long been considered an essential taste in prepared dishes. Umami receptors are similar to the sugar receptors. They are a different kind of GPCR. The umami-specific receptor responds to a specific amino acid, called glutamate.
Glutamate is one of the 20 building blocks that go into making protein. Hence why meats, fish, and nuts often have a savoury flavour to them- they are very rich in protein. Glutamate is also a result of the fermentation process, which is why cheeses and soy sauce are also umami.
Umami by itself tends to be unpleasant (imagine drinking a cup of soy sauce…) but when combined with other flavours generally makes for a hearty and enjoyable dining experience. For centuries, chefs have used broths and sauces to add umami to their preparations and create memorable, unique meals.
Conclusion

Every time you sit down to enjoy a meal, there is a bunch of science going on that determines how well you actually enjoy it. The taste buds in your tongue send signals to your brain, making it act like a portable biochemical characterization instrument, even if we don’t think of flavors that way! Imagine sitting down at a Mexican restaurant- there is the acidity of the lime juice, the saltiness of your margarita rim, the bitterness of cilantro (assuming you don’t have a mutant receptor that just makes it taste like soap!), the sweetness of the tomatoes, and the umami of your carne asada. Every meal can be a scientific experiment!
Now, if you’ll excuse me, I’ve made myself hungry.
Dr. Stefanie Kall is a recent PhD graduate now working as an applications specialist for NanoTemper Technologies. She lives in Chicago, where she conducts extensive scientific experiments in food tasting.