On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...
We’ve all come in contact with pharmaceutical drugs in some form or another. Maybe you take a prescription drug every day to manage a certain health condition. Perhaps you keep a bottle of Tylenol in your desk for when you feel a headache coming on at work. Or maybe you’ve seen advertisements on TV for commercial drugs, featuring people relaxing in bathtubs in an open field or a drug’s name sung to the tune of the Jackson 5’s “ABC.” But have you ever wondered how scientists discovered the exact molecule in the little orange bottle, in your desk drawer or on your TV screen, that does just what it’s supposed to? To understand the process of drug discovery, let’s follow one drug’s journey from the lab to the pharmacy shelf.
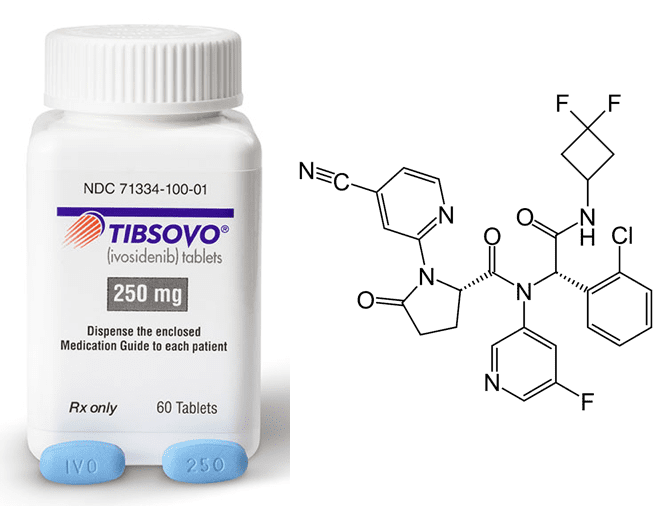
The protagonist of our story is Tibsovo, a drug that the FDA approved last year as a treatment for a type of cancer called acute myeloid leukemia, or AML. Researchers discovered that in AML patients, a certain enzyme—a protein in the cell that helps to convert one molecule to another—was producing a different molecule than usual. By making the wrong molecule, this enzyme set off a chain of events that caused the cells to grow and divide uncontrollably, forming a cancerous tumor. Scientists hoped that by preventing this enzyme from functioning, they could stop the cancer. They therefore aimed to identify an inhibitor for the enzyme, or a molecule that binds to the enzyme and disrupts its function. Two researchers, Janeta Popovici-Muller and Katharine Yen, spearheaded the search for an enzyme inhibitor to develop into a drug for AML.
But how did Drs. Popovici-Muller and Yen start the process of discovering this magical molecule? Finding a molecule that does what you want it to is kind of like winning the lottery. If you were trying to win the lottery, you would buy as many tickets as possible to maximize your chance of possessing the winning ticket. Similarly, scientists test as many molecules as possible—sometimes as many as 3 million—in the first stage of drug discovery to increase the likelihood of identifying one that’s effective.
To evaluate so many molecules in a timely manner, researchers rely on high-throughput screening methods that can process many samples at the same time. These methods often involve plastic containers called well plates that can hold hundreds or thousands of individual samples. Robots help to prepare the samples efficiently by automatically depositing substances like the faulty enzyme and the candidate enzyme inhibitor molecules into the appropriate wells. After the molecules and the enzyme have time to interact, scientists analyze the samples using instruments that can either collect data from each sample in only a few seconds or collect data from all the samples simultaneously. They then use advanced computer software to quickly scan the data and point out samples that showed the desired result. In the search for Tibsovo, the researchers used these high-throughput screening methods to evaluate the effect of millions of molecules on the defective enzyme and found a handful of molecules that inhibited the enzyme. But at this point, they only had the first couple of lottery-winning numbers.
While the high-throughput screen identified molecules that inhibited the enzyme when it was all by itself, Drs. Popovici-Muller and Yen had no idea if they would work when the enzyme was inside a cell. They needed a molecule that could cross the cell membrane to get inside the cell and dissolve so it could travel within the fluids in the cell. Based on its chemical structure, they selected a molecule from their list that was most likely to be able to reach the enzyme in the environment of a cell. They then created about 20 different versions of that molecule and tested them against the enzyme to see which features of the molecule were necessary for it to be effective.
Once the researchers determined the parts of the molecule that were critical for its activity, they designed another set of 20 molecules in which the remaining regions were changed to further enhance its ability to cross the cell membrane and dissolve. They tested these molecules in AML cells in a flask and found one molecule that effectively inhibited the enzyme. They had the next few winning numbers, but they still had a few to go.
While finding a molecule that worked in cells grown in a flask was a major breakthrough, Drs. Popovici-Muller and Yen still had to make sure that the molecule did its job inside a living organism. To do so, they tested their molecule inside a mouse that had been implanted with an AML tumor. Unfortunately, they found that the mouse’s liver broke down and cleared the molecule from the body before it had time to inhibit the enzyme.
They continued to adjust the molecule’s structure to improve its stability inside the body, creating about 15 more molecular variants in the process. One of these variants, compound AG-120, successfully inhibited the enzyme when it was administered to a tumor-implanted mouse. Based on these promising results, AG-120 moved forward into clinical trials, where researchers evaluated whether it was safe and effective for treating AML in humans. Upon taking compound AG-120, a significant number of the trial volunteers entered remission. On May 2, 2019, the molecule was approved by the FDA as a drug for AML under the name Tibsovo. Finally, Drs. Popovici-Muller and Yen were holding the winning lottery ticket!
The average amount of time needed to develop a drug is 12 years, and the story of Tibsovo helps to explain why. While finding one molecule that can overcome so many obstacles will probably always take a long time, scientists are working on making drug development more efficient.
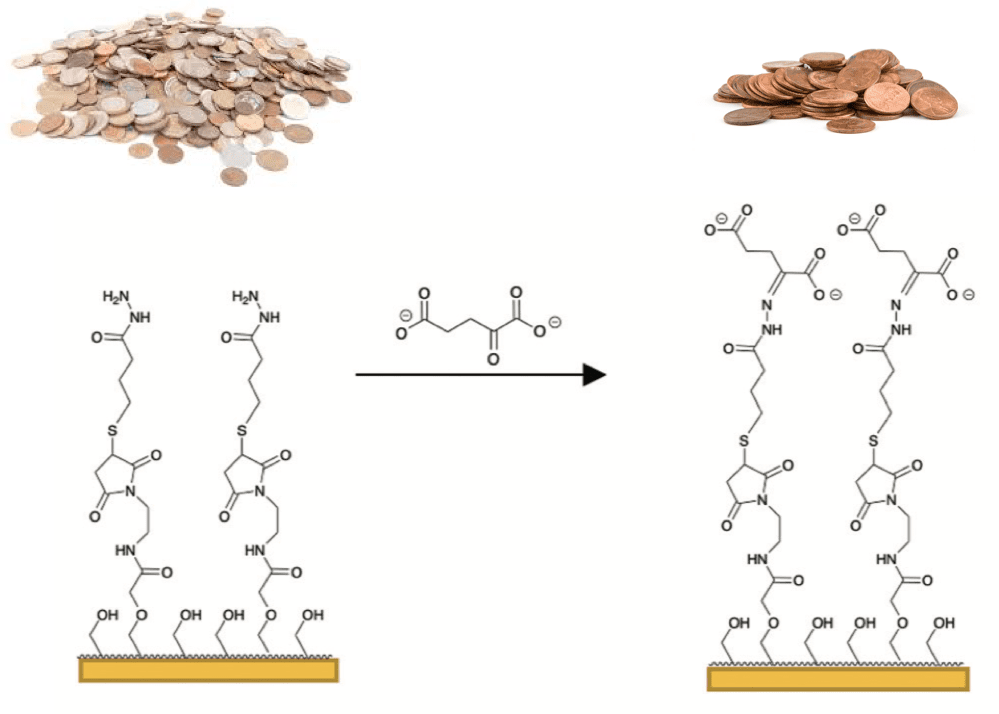
One way to do this is to accelerate the process of testing inhibitors in cells. To see if a molecule inhibits an enzyme, scientists measure the amount of the molecule that the enzyme produces—the lower the amount, the more effective the inhibitor. When evaluating if an inhibitor works inside a cell, researchers use an instrument that separates out all the different molecules inside the cell to isolate and measure the molecule produced by the enzyme. This is a very slow process—it’s like if someone asked you to count only the pennies in a pile of coins (maybe from your lotto win!), so you separated it into piles of each type of coin and then counted the number of pennies.
In my lab, we are developing strategies to rapidly isolate the molecule produced by a specific enzyme. In a recent study, we used a selective chemical reaction to capture only the molecule we’re interested in measuring from a cell. This is much quicker—it’s like if you had a special magnet that attracts only the pennies from the pile so you could isolate and count them more easily.
Other researchers are working on optimizing different steps in the drug discovery process to try to get drugs to the people who need them faster. While we’re at it, maybe drug companies can look for areas of improvement in their advertising campaigns, too!
Sarah Anderson is a PhD candidate in the chemistry department at Northwestern University.