On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...

“I PURPOSE, in return for the honor you do us by coming to see what are our proceedings here, to bring before you, in the course of these lectures, the Chemical History of a Candle.”
-Michael Faraday

In 1860, these words began a series of six lectures by one of the world’s most preeminent physicists, Michael Faraday, on the science of candles. Besides being known as as one of the most gifted scientists in history, Faraday was also lauded for his remarkable ability to communicate science to the public at large. Faraday lived in a time where science communication was not a priority — scientists made their discoveries in laboratories, but they did not see a purpose in educating the public on their matters. Subsequently, in his lecture series, “The Chemical History of a Candle,” Faraday shared knowledge that, until then, was only understood in detail by a handful of scientists and scholars. Faraday’s lectures were so scientifically accurate, and his delivery so clear, that we can still learn from them over 150 years later.
Faraday chose candles as his subject because he understood the scientific richness held within them.
“There is not a law under which any part of this universe is governed which does not come into play and is touched upon in these phenomena. There is no better, there is no more open door by which you can enter into the study of natural philosophy than by considering the physical phenomena of a candle.”
-Michael Faraday
The candle’s function relies on principles from biology, chemistry, and physics. Here, using Faraday as my inspiration, I will explain the science of candles, from its manufacture to its lighting to its flame’s ultimate demise.
The Parts of a Candle

The candle’s structure is rather simple: it is made of wax and a wick.
All candle wax is composed of hydrocarbons, which come from fats. Hydrocarbons are perfect molecules for candles because they liquefy very easily and they store a lot of energy. Europeans throughout history, from Ancient Roman times through the 19th century, used tallow (rendered beef fat) to make candle wax. The ancient Chinese used whale fat, as well as beeswax (which is also rich in hydrocarbons). In India, they boiled down the fruit of the cinnamon tree for their wax, and the Japanese used the extracts of the tree nut. Nowadays, most candle manufacturers use paraffin wax, which is derived from natural oils and carbon-based minerals.
The wick is, and has always been, made of braided cotton because of cotton’s ability to absorb liquids (such as liquid wax.)
Lighting a Candle
When you light a candle wick, you’ve started complex chemical reaction that will keep itself alive as long as the candle is intact and the flame is fed. First, the heat from the flame melts the wax nearby. The wick absorbs the liquid wax, and it travels up the wick to the flame, where it combusts, keeping the flame going.
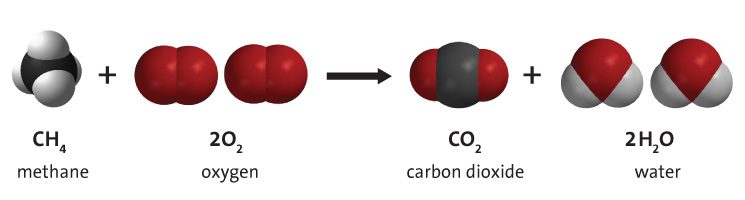
Combustion is a dance. In a combustion reaction, a fuel such as a hydrocarbon dances with oxygen to produce a lot of heat and light.
In a candle, the hydrocarbon fuel comes from the liquid candle wax. As the liquid wax rises up the wick and meets the heat of the flame, it starts to break up into a gas composed of carbon and hydrogen atoms. With their bond broken, the carbon and hydrogen switch dance partners and bond with the oxygen in the air. The carbon’s partnership with oxygen creates carbon dioxide, and water’s meet-up with oxygen makes water vapor.
Some heat from this reaction escapes from the candle flame and keeps us warm, while the rest melts the next layer of wax to continue the chemical reaction downwards.

Parts of the Flame
If you look closely at a candle flame, you’ll notice layers of different color. At the very bottom, near the wick, you’ll find a blue layer. This is the epicenter of combustion, where the hydrocarbons of the wax first meet oxygen and start to break down. Next, you have the dark brown/orange layer, which is where you’ll find some solid carbon. This carbon never had the chance to dance with oxygen, which means it never combusted. Finally, there is the gaseous carbon that escaped the first two layers and combusts just before it can escape for good. This gives the candle it top yellow/white layer. The outermost layer of the flame is the only part that directly comes in contact with oxygen, so it is the hottest layer — it is 2500 degrees Fahrenheit, hot enough to melt steel!
Putting out the Flame
As I mentioned before, combustion requires three ingredients: fuel, heat, and oxygen. In a candle, the oxygen comes from the air, the fuel comes from the candle wax, and the heat comes from a match at first, and ultimately the candle flame itself. As long as a candle has those three ingredients, it will keep burning.
To put out a candle flame, all you have to do is take away one of these three ingredients. The simplest option is to let the candle run out fuel on its own — let it burn to its base and run out of wax. Another option is to blow out the flame. Blowing on a flame cools down the air around the wick, which removes the heat. Finally, you can burn out a candle by depriving it of oxygen. You can do that really easily if your candle comes in a jar — just place the lid on the jar, and the candle will blow itself out!
Candle Smoke
When you blow out a candle, why does smoke start billowing off the tip of the wick? When you blow out a flame, you take away the heat, which prevents the liquid wax from turning into a gas in the outer portion of the flame before it combusts. Consequently, the hydrocarbons get stuck in liquid form and cool back down into solid particulates. These particulates simply float away in the form of smoke.
Sometimes, candles leave behind a thick layer of dark black soot, which is like a water spot on a drinking glass. Smoke is composed of millions of microscopic droplets (instead of water, they are made of carbon). When smoke lands on a cool surface and “dries,” it leaves behind soot.
The Study of Candles
Candles have been used for thousands of years, and for most of those years, the makers and users of candles had no idea how candles work. Nonetheless, they were fascinating enough to attract Michael Faraday’s attention, and even 150 years after that, candles still carry a somewhat mystical aura. Faraday’s lecture series on candles was part of an annual lecture series he started in 1825 at the Royal Institution in London to bring science education to the general public, especially younger people. The lecture series, called The Royal Institution Christmas Lectures, continues to this day. You can watch the lectures here.