On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...
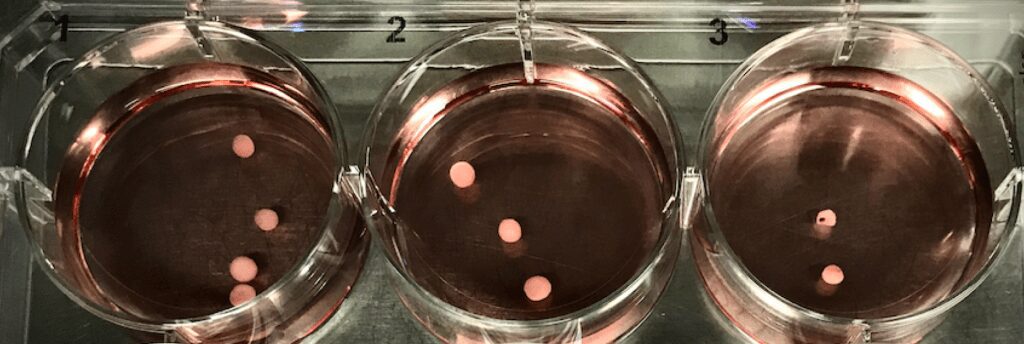
Mini-brains—that’s right. It sounds like something from Frankenstein, doesn’t it?
I gazed in awe as my mentor showed me these ‘mini-brains,’ aka cerebral organoids. Much like soufflés, mini-brains must grow without falling apart and require a lot of care and patience. They ‘rise’ to the challenge of helping physicians and scientists better understand the brain and develop treatments for diseases that affect us and our loved ones.
Mini-brains are not exactly “miniature brains” as the name implies; rather, they are 3D collections of brain cells – like a blob of cells all stuck together. Mini-brains can be created by taking blood cells from patients and changing them into special cells, called ‘stem cells’, that can mature into almost any kind of cell. In other words, a mini-brain is basically a bunch of different cells all stuck together interacting with each other. Being able to use cells like this means we don’t always have to count on animals, like mice and fruit flies, to study our diseases. Sometimes we can use stem cells to learn about diseases instead. One limit to this technique is that mini-brains don’t have the structure that animal brains have. However, they do mimic and help us understand how different human brain regions develop and interact, especially at early stages!

For example, mini-brains can help doctors understand how developmental delay occurs in infants. To grasp how mini-brains rise to this occasion, let’s consider the story of Sophie, a girl born with a delay in brain development, epilepsy, and permanent neonatal diabetes, a type of diabetes that affects babies in the first six months of life and lasts for their entire lives. The combo of these three symptoms is known as DEND Syndrome. Although DEND Syndrome affects one in a million, it could affect any baby, just due to a change in a single gene.

At 4 months old, Sophie had sky-high blood sugar and she was misdiagnosed with Type 1 diabetes, which meant she would have to be treated with insulin every few hours. Sophie had a hard time smiling, struggled to hold her head up, and couldn’t latch onto her mom to feed. When she turned 3 years old, she finally had genetic testing done, and scientists found a permanent change, aka mutation, in a gene that helps control blood sugar and affects how brain cells communicate. This gene is called KCNJ11.
Once the mutation was found in Sophie, her doctors started treating her with medicine that brought her blood sugar down and helped improve her development. When Sophie turned 5, she still wasn’t walking or talking, but by age 11, Sophie could walk, smile, and interact with others. If doctors had started treatment much earlier, Sophie might not have experienced as much developmental delay. So the question arises: Just how much earlier should her doctors have started treatment? Could they have treated her while she was in the womb itself?
That’s where mini-brains come into the picture.
In the Kovler Diabetes Center, where I work as a Student Research Assistant, we’ve taken cells from patients with the same mutation that Sophie has, to start answering these questions. We create mini-brains using these cells and compare mini-brains with and without the mutation. Studying these mini-brains helps us understand how the developmental delay begins at stages when we don’t otherwise have access to human brain samples. Similar to how we bake and decorate soufflés for our culinary delight, we grow, preserve, and cut mini-brains into slices thinner than a human hair! We then ‘paint’ the mini-brains using proteins, called antibodies, that can find and attach to other proteins, which helps us see where they are located in the mini-brain slice. We are also treating the mini-brains with the same medicine that was given to Sophie to improve the development of the mini-brains carrying the mutation. From our results, we will aim to figure out the stage of development when it is most logical to start treatment and personalize medicine for patients like Sophie.
Mini-brains also help us understand which treatments work best for patients with brain diseases. At Weill Cornell Medicine, a brain cancer specialist has started making mini-brains that also have tumor cells from patients. By the end of 2019, he hopes to start the first clinical trial involving mini-brains by testing which treatments can best fight each patient’s tumor. Finding out which treatments do and don’t work for each patient saves a lot of time and money. This personalizes your medicine, much like eating the perfect soufflé made for your taste buds instead of trying a bunch of soufflés that don’t taste as good.
Mini-brains rising into space

In addition to tailoring our medicine, mini-brains help us look ahead to our possible future as a species dwelling on multiple planets. The first question we need to address is whether we can even survive and develop normally in space—and mini-brains will take us closer to getting an answer. Just like how soufflés seem to defy gravity with their airy appearance, mini-brains have also defied gravity and been launched to the International Space Station. In space, the mini-brains’ cells interact differently, which means we can study how brain diseases occur in ways that are impossible on Earth. Living on other planets as of now might seem far-fetched (or maybe not if you’re a Star Wars fan). But with climate change and the promise of space exploration, it’s a reasonable line of research for our survival in the long-run.
Mini-brains can rise up to many challenges, but will they?
Stem-cell research helps us understand disease and personalize medicine. But without funding, the research won’t continue to happen, and our metaphorical soufflé will fall flat. For mini-brains to rise up to the challenge, researchers need your support. Donate to the National Stem Cell Foundation, and reach out to legislators. And, last but not least, share this article with your friends and family to help mini-brains rise to their fullest potential like a good ol’ soufflé!
Note: Names of patients have been changed for their privacy.
Swathi Balaji is a 4th year undergraduate at the University of Chicago, majoring in Biology and minoring in Health and Society. She plans to pursue a career in medicine, and she loves to sing, dance, and create comics in her spare time. You can find her on LinkedIn.