On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...
The extent of the average American’s knowledge regarding mitochondria is that seemingly-ubiquitous adage from high school biology class: mitochondria are the powerhouse of the cell. But not everyone knows just how true that is, or the many fascinating biological secrets packed into these tiny organelles.
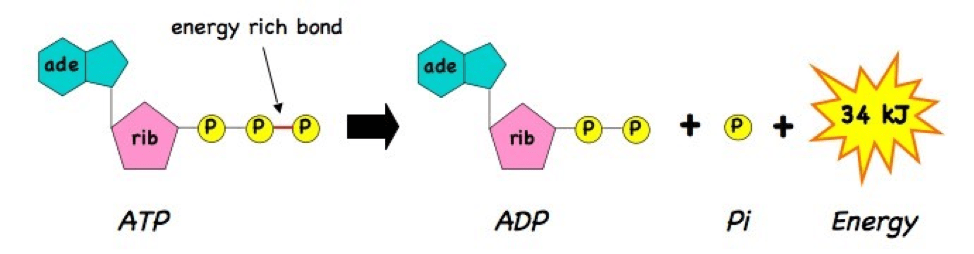
Many important reactions in the cell need to harness the energy stored in the bonds of a molecule called adenosine triphosphate (ATP), which contains three chemical groups called phosphates. ATP is the energy currency of the cell — in fact, life as we know it cannot exist without the breakdown of ATP. ATP powers reactions when it loses one of its phosphate groups, forming adenosine diphosphate (ADP). That third phosphate has to get reattached to ADP in order for ATP to be used again, and that is the main purpose of the mitochondria.

Mitochondria earn their powerhouse moniker because they are really efficient at making ATP. Efficiency refers to the portion of a fuel’s stored energy that a system uses, while the rest is lost as heat. Different systems use different fuel sources – for example, most automobile engines use gasoline, while mitochondria rely on food like sugars and fats. Mitochondria break down the bonds in sugar and fat molecules to release energy. They then use this energy to power a series of reactions that reattaches the third phosphate group to ADP, regenerating ATP as a source of energy the cell can use. Most car engines are between 20 and 30% efficient in extracting energy from gasoline to make a car move, while mitochondria harness about 40% of the energy from food molecules to produce ATP. The 60% lost as heat may seem like a big waste, but this is what maks our bodies warm, so even the “wasted” energy serves a purpose.
Cells with mitochondria are bestowed an incredibly powerful advantage. With such an abundance of usable energy, cells can make more proteins, get larger, move further, and communicate with each other. Some evolutionary biologists theorize that mitochondria are the reason that cells were able to start organizing into multicellular organisms, such as you and me.
According to the endosymbiotic theory of life, there was once a time millions of years ago when primitive eukaryotes (cells that contain a compartment for DNA called the nucleus) hadn’t yet acquired mitochondria. Eventually, some eukaryotes likely tried to eat mitochondria-like bacteria, failed to digest them, and ended up with new mitochondrial compartments. The eukaryote benefits from the immense amount of ATP energy produced by mitochondria, and the mitochondria enjoy access to nutrients and shelter from their hosts.
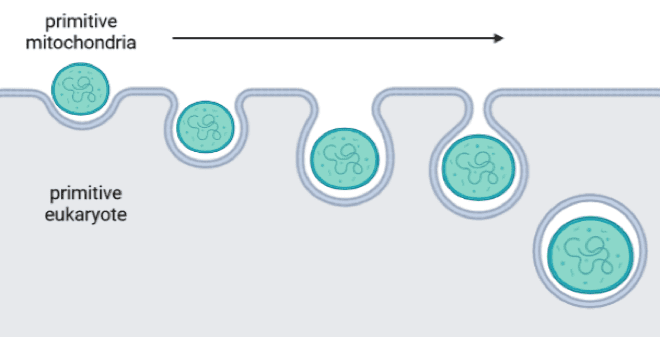
A few intriguing traits of mitochondria serve as the pillars of the endosymbiotic theory. First, mitochondria are the only animal cell compartments that have their own DNA (called mtDNA). Several genes in mtDNA serve as the blueprint for proteins within the mitochondria that are required for ATP generation. In humans, the DNA in the nucleus comes from both parents, while mtDNA is passed down solely from the mitochondria packed into the egg. Single-celled organisms like bacteria similarly inherit all their genes from only one parent during replication, supporting the idea that mitochondria originated from bacteria.
Another piece of evidence for the endosymbiotic theory is the presence of double membranes surrounding mitochondria. Picture the cell as a magic water balloon that can pinch off parts of its outer latex without popping. Imagine gently pressing your index finger and thumb into the balloon, and then pinching off the pocket you made so that another mini balloon is now floating inside the water of the first. Other cellular compartments were formed this way – they have only one membrane, suggesting that the cell membrane folded in on itself to make little pockets. Now imagine that as you approach the water balloon, you press another, tiny water balloon against its surface and pinch off a pocket so that the tiny balloon is totally enveloped in latex from the big balloon. Unlike the first floating mini balloon, this one has two layers of latex (see figure of endocytosis process). The fact that mitochondria have two layers of membrane indicates that they already had their own membrane when the eukaryotic cell membrane folded around it. Since bacteria have a single membrane, it would make sense that mitochondria started as ingested bacteria.
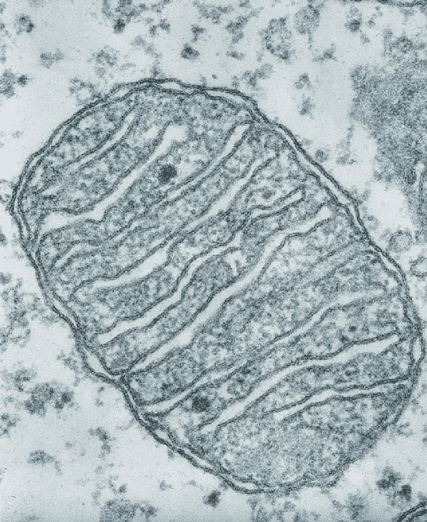
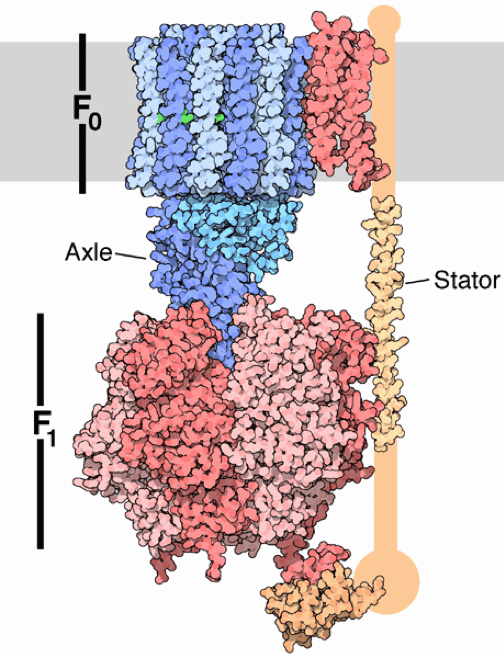
The two membranes are critical to the function of modern mitochondria, with different proteins embedded in each membrane and electrically charged atoms called ions stored in the space between. One important protein located on the inner mitochondrial membrane is ATP synthase, which carries out the final step of the series of reactions that create ATP. During the first steps of the process, hydrogen ions build up between the two membranes like water behind a dam and flow through the F0 region of ATP synthase (see figure of ATP synthase) like water turning a miller’s wheel. This motion causes the F1 domain, holding ADP and P, to change shape and smash them together to make ATP. The protein then releases ATP so it can be used by the cell. You can see a video representation of this remarkable reaction here.
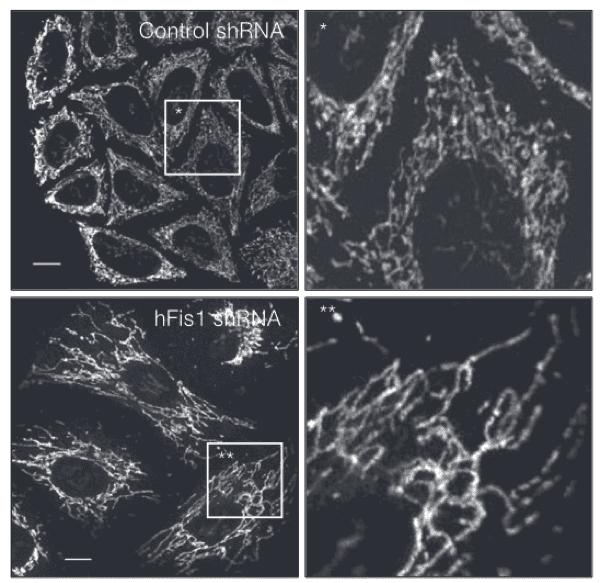
Even though mitochondria are clearly great at what they do in the realm of energy production, biomedical research is discovering additional roles for mitochondria in the cell. Mitochondria have been found to sequester certain proteins, regulating a cell’s response to stress. They can interact with other cellular compartments to mediate nutrient balance and signaling within the cell. The vast mitochondrial network undergoes constant remodeling: individual mitochondria fuse and split from one another, and parts of the network are broken down when they’re damaged or when the cell doesn’t need them. Mitochondrial network remodeling is particularly relevant to cancer, since tumor cells alter the production and use of energy so they can survive and divide out of control.
Ultimately, mitochondria are more than just the powerhouse of the cell, though their supreme energy efficiency has certainly earned them that particular medal. They are also hosts to some of the most awe-inspiring clues to our evolutionary origins, amazing molecular machinery that is equal parts powerful and elegant, and avenues for superior understanding of disease.