On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...
In 2013, when Tenzin Kunor was in his last semester of college, he learned that he had a type of tuberculosis (TB) that was resistant to the typical drugs that are used to treat TB. At first, he thought it was a bad cold because he had been experiencing chest pains, a sore throat, and coughing. When it got to the point that it was painful to walk, he received his diagnosis: multidrug resistant TB. This bacterial infection requires special medications, and takes a long time to resolve. For Tenzin, it took two years.
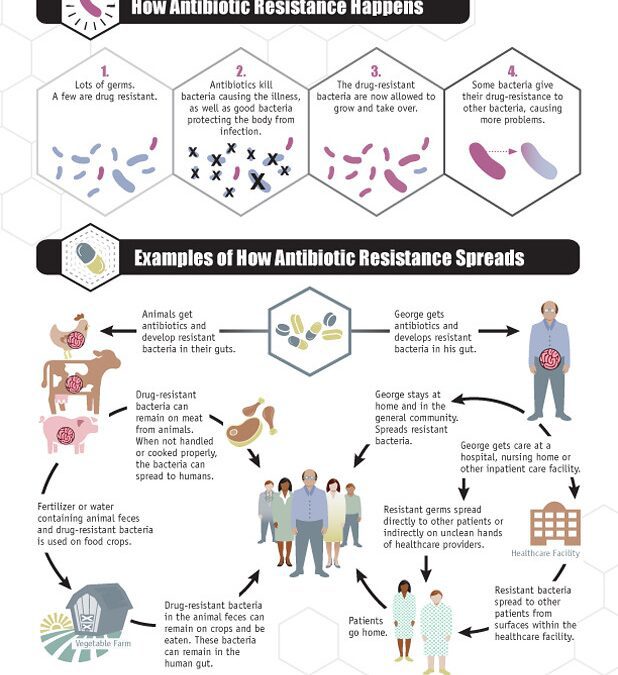
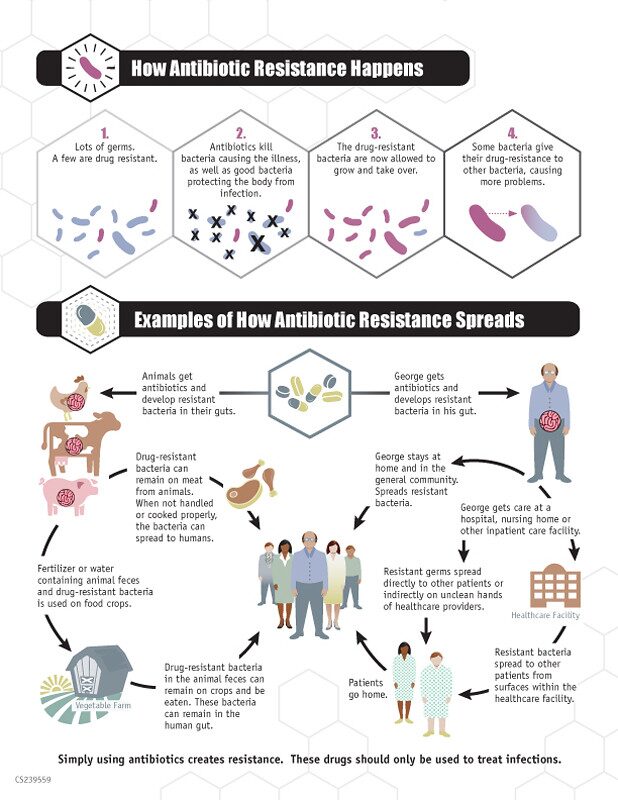
Unfortunately, Tenzin’s story is not uncommon. According to the World Health Organization, antibiotic resistance is one of the biggest threats to global health, development, and food security. In the U.S. alone, at least 2 million people are infected by antibiotic-resistant bacteria and about 23,000 people die per year. The rapid increase in these bacteria is due to the overuse of antibiotics, as well as the low rate that new antibiotics get developed.
Bacteria can become antibiotic resistant in different ways: by breaking down the antibiotics, or by pumping antibiotics out of themselves, or by changing their cell structure so that the antibiotics cannot enter. Alarmingly, bacteria can also pass these defense mechanisms to other antibiotic-sensitive bacteria by sharing their DNA, creating an army of infections that are difficult to treat. Tenzin required over 8,000 pills before he was considered cured.
The Enemy of My Enemy
Researchers are now trying to defeat antibiotic-resistant bacteria by teaming up with bacteria’s natural enemy: bacteriophages. Bacteriophages are viruses that exclusively target bacteria and kill them. Bacteriophage therapy offers a unique advantage because they have been locked in an evolutionary battle with bacteria for millennia. Every time bacteria evolve to prevent bacteriophage infections, viruses evolve a new way to kill them.
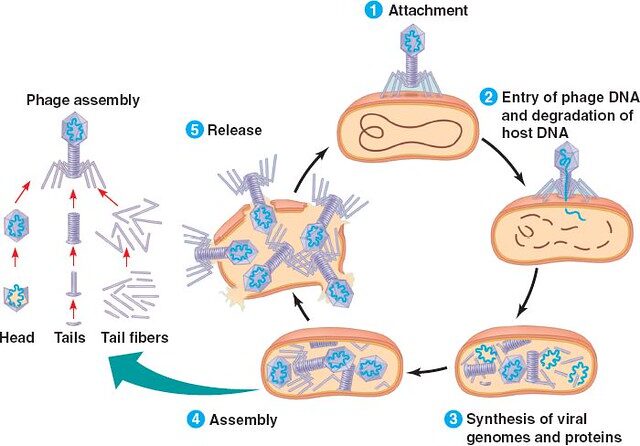
A team of researchers from the University of Pittsburgh has recently engineered bacteriophages to treat a 15-year old cystic fibrosis patient who was infected with Mycobacterium absessus, which is a close relative of Mycobacterium tuberculosis, and is frequently found in patients with cystic fibrosis. Cystic fibrosis patients often need a lung transplant, but they have higher mortality rates during surgery because of the persistent infections that are present prior to the surgery. M. absessus is particularly difficult to manage because of its ability to resist antibiotic treatment.
Shaken, Not Stirred
The Pittsburgh team dove into a collection of more than 10,000 bacteriophages that had been isolated by students. They modified three bacteriophages (Muddy, ZoeJ, and BPs) by changing around the order of some of their amino acids to make the most efficient mixture of phages that can kill the bacteria. Then, they administered this three-phage cocktail to the patient. All three phages look similar, with a round head attached to a cylindrical tail. However, they carry different genetic material which can kill M. absessus, presumably by targeting the bacteria in three different ways. This combination makes it less likely for M. absessus to develop resistance to the cocktail.
Doctors started this treatment after the Pittsburgh cystic fibrosis patient received a lung transplant. Prior to the transplant, the patient had a chronic M. absessus infection and was on antibiotic treatment for 8 years. But after the surgery, the antibiotics caused bad side effects, necessitating the development of the viral cocktail. The new treatment was effective in reducing the symptoms of the bacterial infection within a month. Over the next six months, the patient’s lung function also improved. Usually, patients in these situations do not survive, giving hope that the treatment will help more patients in the future.
Unfortunately, there are some aspects of the treatment that still need to be resolved. The susceptibility of M. absessus to these viral cocktails differs based on the patient. Therefore, the cocktail will need to be personalized, setting up a race between the researchers and the bacteria. Another patient who was part of the study died before the researchers could find the right viral mix. Luckily, the researchers have and will continue to save many patients through the optimal unique mixing of phages. For Tenzin, the individual quarantined with tuberculosis, it took years of special medications to overcome the multidrug resistant disease. For all patients with drug resistant infections, creating a compendium of all the possible phages that can kill the infection appears to be the future of quick and efficient treatment.