On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...
Has your dentist ever warned you that “acidic” drinks are bad for your teeth? Have you ever heard a personal hygiene product advertised as “neutralizing”? These terms, while used somewhat frequently in everyday language, actually refer to a scientific concept called “power of hydrogen,” or pH.
pH is a numerical value that is measured on a scale from 0 to 14. It describes a substance’s tendency to give or receive protons, which are positively-charged hydrogen atoms. Materials with a pH below 7 (such as orange juice) like to give away protons and are classified as acidic, those with a pH above 7 (like soap) prefer to take up protons and are considered basic, and those with a pH of 7 (such as water) are called neutral and neither give nor receive protons. The lower the pH of a substance, the more acidic it is and the more willing it is to give off protons. The higher the pH of a substance, the more basic it is and the more likely it is to take up protons.
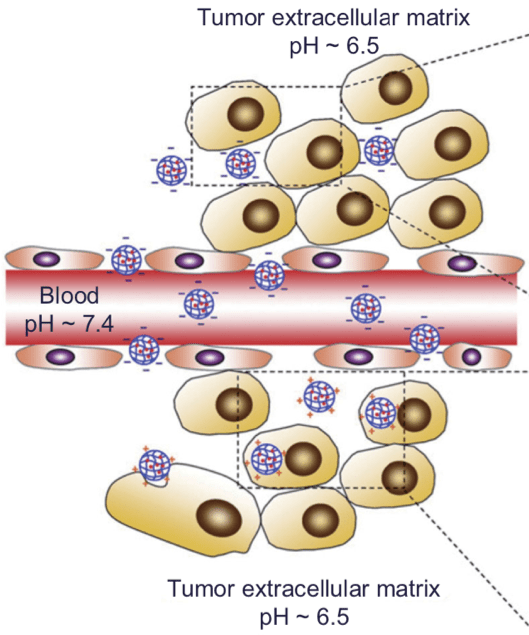
As it turns out, pH can impact our bodies in ways that are more significant than degrading our tooth enamel. In fact, pH plays a critical role in cancer. Cancer cells rely on different chemical reactions than healthy cells to make the molecules they need to grow and divide at a fast rate. This unique set of reactions also produces a large quantity of protons, which are then transported from inside to outside the cell. This abundance of excreted protons gives cancer cells an acidic environment: the pH at the surface of a cancerous tumor typically ranges from 6.5-6.8, while the pH inside the rest of our bodies is about 7.4.

The relationship between cancer and pH is cyclical: while cancerous cells produce an acidic environment, this acidic environment, in turn, contributes to the progression of cancer. Scientists believe that the acidity surrounding a tumor kills nearby healthy cells, creating space for cancer cells to invade and allowing the tumor to grow. The abnormal pH around tumors presents a new angle into cancer therapy, and two different approaches have emerged that use pH to treat cancer.
First, some researchers are developing strategies to slow tumor growth by increasing the pH outside of cancer cells to match the pH of the rest of the body. Increasing the pH surrounding cancer cells requires reducing the number of protons outside the cell. One way to do this is to treat the tumor with something that accepts protons: a base! Researchers at the Moffitt Cancer Center used sodium bicarbonate (also known as baking soda, another common household base) to soak up the protons excreted by cancer cells like a sponge. They implanted mice with colon tumors and divided them into two groups, one of which drank regular water and one of which drank a sodium bicarbonate solution. After the treatment period, the researchers measured the pH at the tumor’s surface and the volume of the tumor. They observed that in the mice that drank the sodium bicarbonate solution, the pH of the tumor’s environment was closer to 7.4 and the tumor was smaller compared to the mice that drank the water. These results demonstrate that the sodium bicarbonate successfully adsorbed the excreted protons, increasing the pH around the tumor and slowing its growth. While this study is an interesting demonstration of consumption of an edible base for cancer therapy, this treatment method has not yet been validated in humans. Check out my companion article to learn more about the relationship between the pH of your diet and cancer.
Another way to decrease the number of protons outside cancer cells is to prevent the cells from being able to excrete them in the first place. Cells transport protons across the cell membrane using proton pumps, which are like little tunnels through which only protons can fit. Researchers have developed molecules called “proton pump inhibitors” that change the conformation of these tunnels, causing them to become closed off and keeping protons from passing through. Proton pump inhibitors are commonly used to treat acid reflux by stopping the cells in the stomach lining from secreting protons to produce stomach acid. However, scientists are currently exploring the application of proton pump inhibitors toward cancer therapy.
Researchers discovered one proton pump inhibitor that prevented liver cancer cells from excreting protons, increasing the pH surrounding the cells and decreasing the rate at which the cells multiplied. The drug, called cariporide, is currently in clinical trials to treat a variety of cancers.
Rather than trying to treat cancer by neutralizing the pH of cancer cells, some efforts are focused on using the acidic conditions of the tumor environment to improve existing therapies. One of the major goals in cancer research is to develop treatments that are more effective and cause fewer side effects because they can specifically interact with cancer cells over healthy cells. With this aim in mind, scientists have harnessed the unique pH of the tumor environment to selectively deliver drugs to cancer cells. The drug can be hidden in a larger structure through chemical bonds that are strong at the normal physiological pH, but fall apart at an acidic pH. Therefore, as the structure circulates the body, the bonds remain intact and the drug stays hidden. However, when the structure enters the acidic environment of the tumor, the bonds break, releasing the drug. In this way, the drug compound is delivered to cancerous cells, but not to healthy cells.
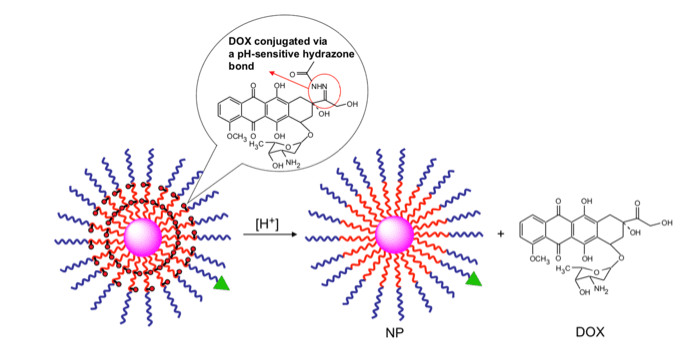
In one example, researchers attached a chemotherapy drug called doxorubicin to the surface of tiny gold particles using chemical bonds that break under acidic conditions. They treated cancer-infected mice with the complex and observed that the drug was delivered only to the site of the tumor. In another example, scientists encapsulated the drug camptothecin into a cage-like structure made up of metal atoms connected by molecules. The bonds between the metal atoms and the molecules were designed to break at an acidic pH, causing the “cage” to degrade and release the compound in the presence of cancer cells. This structure was shown to selectively interact with and kill breast cancer cells.
Emerging pH-based cancer treatments—whether they directly target the acidic tumor environment or indirectly use its distinct pH gradient to maximize the potency of other drugs—hold much promise in the field of cancer research. While increasing the pH surrounding cancer cells only slows tumor growth rates, acid-neutralizing methods could be administered in combination with other drugs to more effectively treat cancer. pH-responsive molecular structures can selectively deliver drugs with established therapeutic activity to cancer cells, but still need to be evaluated in a clinical setting. Scientists plan to further investigate these existing treatments and to explore new ways in which pH can be exploited to fight cancer. But regardless of what the future holds, it’s clear that the power of hydrogen is undeniable.

Sarah Anderson is a PhD candidate in the chemistry department at Northwestern University.