On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...

The use of animals in research has been a controversial topic for some time. There is still a divided opinion on animal-based research, with many people believing that animals are an ideal choice to not only enhance our understanding of complex diseases such as cancer, but to also test drugs and cosmetics. However, many people argue that using animals is unethical, cruel, and unnecessary.
In Great Britain alone, the use of animals increased by 6% in 2021. This is likely to rise as diseases are becoming more complex and the demand for ground-breaking research is constantly growing. In an ideal world, we wouldn’t have to harm animals at all for research purposes, so what other options do we currently have?
Growing cells in 3D is really, really tricky…
Human derived ‘cell lines’ are usually the easiest and most reliable option for studying human diseases or testing new drugs. They are originally derived from healthy or unhealthy tissue samples donated by consenting patients, where they are then frozen, stored, and reused over and over again for experiments. Whilst they provide a living model to experiment on, they are still far from ideal when investigating complex diseases or drug interactions. Cell lines only ever grow in a 2D layer at the bottom of a petri dish, never in 3D like an actual living organ. Because of this, they also grow in a strange arrangement – on top of each other, upside down or back to front, which is not what happens in the body. Another shortfall of these simple models is that only one cell type is growing in the dish at a given moment, meaning that they are incredibly basic, and it is difficult to draw a solid conclusion from them without repeating the same experiments on other types of cells.
Modelling a piece of living tissue or whole organ without resorting to animal work is a difficult task. Over the past decade, engineered, or plant-based scaffolds became popular amongst scientists and researchers, which can be coated in cells and studied like a living organ. Plant based scaffolds are derived from natural substances, which are engineered to interact with and promote the growth of living cells. Many natural materials have been tried and tested, but some of the most interesting ones include alginate (seaweed), starch (plants) and cellulose, which can be harvested from apples. The general idea is that these materials are sterilized, prepared and then cells can be seeded on top of them in a 2D layer. Cells are a lot happier when they can grow on a suitable surface, as opposed to a petri dish! There are several applications for this technology, such as using plant-based scaffolds to generate large numbers of cells which can be used for further experiments, like testing drugs. In addition to laboratory work, there is potential for this scaffold-plus-cell technology to be used in medicine, such as a replacement for skin grafting. Unfortunately, this type of application is still under scrutiny, as many factors need to be considered, such as allergic reaction, inflammation, and rejection. Just because a material behaves a certain way in the laboratory, it may behave totally differently once inside the body and even degrade or cause more harm than good.
If natural cells are becoming too basic to study tricky diseases, even when grown on a scaffold, is it possible to grow a functioning organ instead?

Organoid discovery
This is a particular question which has been on a lot of peoples’ minds for some time, and it turns out the answer is a lot more complex than we think.
At this time, methods to grow fully functioning organs from scratch in the laboratory are yet to be discovered. Instead, ‘organoids’ are becoming popular models in laboratory research. Organoids are a halfway house between cell lines and whole functioning organs; they are very small, round, and originate from induced pluripotent stem cells (iPSCs), embryonic stem cells (ESCs) and even healthy or cancerous patient-derived adult stem cells. But how did scientists figure out how to turn stem cells into organoids?
Dutch molecular geneticist and Keio Medical Science Prize winner Hans Clevers and his team at the Hubrecht Institute were the first people in the world to pioneer organoid research. In 2008, Clevers hypothesized that he could use intestinal biopsy tissue to obtain stem cells, which could then be used to make even more stem cells. What actually happened was ground-breaking. Not only did more stem cells form as he expected, but the bunch of cells in the petri dish resembled the original tissue and started to look like more like the small intestine! This was the birth of the first ever organoid.
Since this discovery, several researchers have been trialling methods developed by Clevers. Both healthy and diseased tissue can be harvested, usually by biopsy, from a variety of consenting patients who then go home after the procedure and continue with their day. This tissue is then transferred to a laboratory, where complex protocols are carried out to retrieve the stem cells and develop them into organoids.
Laboratory grown ‘personalized’ organoids
Organoids are not just limited to the intestine either, as researchers have successfully developed healthy patient-derived organoids from the stomach. These organoids can then be used in the laboratory to model the stomach lining, for example, to simulate stomach ulcers or cancer development. Unhealthy patient tissue, such as tumors, have also been transformed into patient derived tumor organoids (PDTOs), also known as tumoroids, including breast, colorectal, liver, brain, and pancreatic cancers. Tumoroids in particular are a huge breakthrough in cancer research as they allow scientists to investigate and study tumors outside of the body. For example, cell signaling and interactions with anti-cancer or chemotherapy drugs can be observed extensively, without having to experiment on the patient or replicate the disease in animals.
There are many applications and types of research which use organoids in addition to drug discovery and disease modeling. Further applications are shown in the image below.
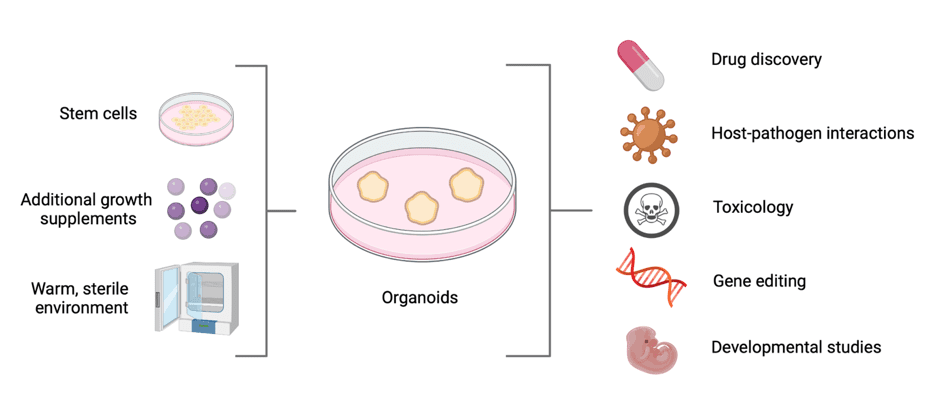
Human derived organoids have opened up a world of opportunities for scientists and researchers alike. They provide an ideal model to carry out a range of experiments, without having to harm an animal. Organoids help to solve the problem associated with 2D cell lines, as they provide a variety of different cell types whilst also resembling the original tissue or organ somewhat. For example, this means that researchers do not have to test the same drug or technique on several different cell lines at any given moment in order to reach a conclusion. Organoids have also opened the door for ‘personalized medicine’– if patient-derived organoids are responding to drugs in the laboratory, does this mean these drugs can be safely given to the patient? We are still far from drawing this conclusion because organoids are still relatively simple systems, but they may replace the need for extensive and expensive clinical trials in the future.
Whilst promising, it is worth mentioning that organoids do lack many features which we take for granted in our bodies, such as a circulatory, respiratory, and immune system. Currently, the majority of organoid research does not consider these factors, which sets a limit on the conclusions that can be made by researchers. Another issue is the size of organoids, as they are usually no bigger than 1mm in diameter. Because they are so minuscule, they are fiddly to work with and require complicated growth requirements and maintenance.
Ultimately, we are still a long way from completely eradicating the use of animals in research, but organoids are an exciting prospect for the future of biological research and are definitely a step in the right direction.