On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...

The year was 2008. A year of recessions, Obama, and the last Batman movie to be filmed in Chicago. It was also a year when a mistake of mine led to a loss that still affects me today: that of my beloved wool trousers from Nepal.
Purchased at a store in Berkeley, these pants were made in Tibet and were everything my little hippie heart wanted in a piece of clothing. Fair trade. Embroidered with little wildflowers at the seams. An odd shade of maroon. Oh, and of course, made of organic yak wool. They were my pride and the envy of the rest of my cohort at UC-Berkeley that summer. Imagine my horror, when I stupidly tossed them in with the rest of my clothes that needed to be washed later on that year. Not thinking, I put them in the same load as my jeans and work out clothes, cranked the setting to ‘High Wash,’ and then threw the clean pile into a subsequently hot dryer.
Blame my ignorance, or my exhaustion in graduate school. The trousers were subsequently shrunken and shredded, even though the rest of my clothes were fine.
Rest in pieces, my beautiful, organic yak wool trousers. Literally.
Why did my washer/dryer have to do this? There, of course, we find some science.
Clothes make the man (or woman, or person, really). How we treat our clothes is crucial to them surviving in the long-term. The daily wear and tear that results in the dryer lint we pull out of the machine after a couple of cycles shortens their life, but putting them on the wrong cycle in the washer or dryer can ruin them even sooner. Taking better care of our clothes causes us to purchase less and keeps our environment in a nicer shape then if we had to constantly consume more products.
Take a look at your tag of your shirt (not now!). What does it say?
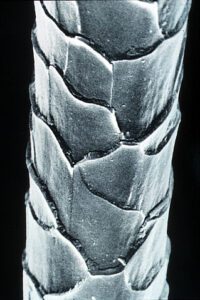
Merino wool: a finer wool with smaller scales, translating into a smoother feel but just as much shrinkage in the laundry!
Is that comfy sweater made of wool, such as Angora? Wool, and other animal products such as silk, needs to be washed gently, on a light cycle. Sure, that gnarly Irish sweater may have helped you survive another South Side Irish parade, but it isn’t as tough as its brillo-like texture would suggest – and that’s because of its structure. Wool is covered with scales, like a snake. As you may recall from my previous post on knitting, wool is pretty elastic, and this snake-like material absorbs a lot of water. When you throw your wool sweater into a hot wash, those scales on the wool open up and stretch out like hooks on velcro, and different wool strands get stuck to each other and lock into place. Such an occurrence of duress for the fabric is called ‘felting,’ and it ends in destroyed clothing. Cotton displays a similar behavior, but the plant-based structure does not produce the legendary wash-reaction that animal blends do. Certain wool blends may be more durable (such as Smartwool), if they’ve been blended with a different type of fabric, which is usually polyester.
Polyester and its cousin, nylon, are tougher than tough, which is why you find this polymer-based fabric in everything from work polos to sleeping bags. Some people turn their noses up at this material because its cheap to manufacture, but it serves a very important purpose. Because of the construction of the fabric and it’s lack of a lipid protein structure (which would repel water) it absorbs sweat from the body better and cools quickly. Its structure also allows a lot of stretch, a crucial engineering feat for active individuals. As a result, polyester can really take a beating in the laundry, and it is okay to put it in hot water and a hot cycle (and it’s probably good after you’ve finished sprinting in that Pi Day run!). Not surprisingly, polymer-based materials really took off during World War II due to the need for easy-to-clean, easy-to-manufacture, and cheap-to-produce synthetic materials. I daresay you’re probably wearing at least one item with this chemical background.
Speaking of synthetic, the type of soap someone puts on their laundry also affects how his or her laundry will turn out in the wash. What did I put on those organic wool trousers of mine? You can bet it was generic laundry detergent with a good helping of bleach (My hiking clothes were stained beyond belief and needed a solid rinse)! Historically, the soap that many cultures have used to clean clothing has been made out of fat and lye. Lye bleaches materials, partly because it can turn into a caustic basic solution when mixed with water, which subsequently breaks up pigment molecules. This allows your whites to stay white, and your maroon trousers to turn a weird pinkish shade. Meanwhile the fat causes the bacteria in your clothing to disintegrate and fall off your clothing. With that being stated, if you really need to get a stain out of a colored piece of clothing, I’d personally recommend another detergent without any bleaching power, or perhaps some good old hand washing.
(I’d also not recommend making polyurethane foam in your favorite clothes, as STEM Scouts at STEM Con at College of DuPage did this year!)
Companies have added chemicals to laundry detergent not only to clean your fabrics, but to also make your clothes smell a little fresher and nicer. Why does laundry smell so bad in the first place? It’s not just sweat you’re smelling. Prof. John Dean from the University of Northumbria in the UK explains that bacteria from your skin, including both the Corynebacterium and Staphylococcus (yes, the frightening Staphylococcus of MRSA fame!), land on our clothes all the time and produce volatile –and pungent — compounds that make our laundry smell nasty.
Even though chemical and fiber science may seem daunting, it’s important to keep our clothes clean. Studies have shown that frequent washing may have saved us during the industrial revolution, when may people died from bacterial infections (Wrigley, 1990). I know my wool pants did not die in vain, but for a greater need to keep us healthy and smelling nice.
Further Reading:
John Dean (2006) Smelly Laundry? It’s All Down to Chemistry
Luz Claudio (2007) Waste Couture: Environmental Impact of the Laundry Industry
E. A. Wrigley (1990). Continuity, chance and change: The character of the industrial revolution in England. Cambridge University Press.