On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...

From diamond rings to sapphire earrings to garnet necklaces, people have gotten into the habit of wearing and cherishing gemstones. Maybe it’s their rich colors, their sharp facets, or the way the light glimmers when it hits them; natural crystals are definitely some of Earth’s most extraordinary materials. Somehow, along with all its craggy rocks and chalky surfaces, nature finds ways to arrange atoms into (nearly) perfect patterns, and the result is the smooth, clear crystals we love to wear. But gems aren’t special just because of their aesthetic appeal. Because of their purity and the way they shine, scientists covet them for use in the laboratory too!
The First Ruby Laser
In 1960, Theodore Maiman invented the first laser – and it featured a ruby. Rubies are basically sapphires, which, in their purest form, are transparent, single crystal aluminum oxide. The composition of rubies only differs by a few chromium ion impurities. These impurities absorb blue and green light, giving rubies their lovely red hue.
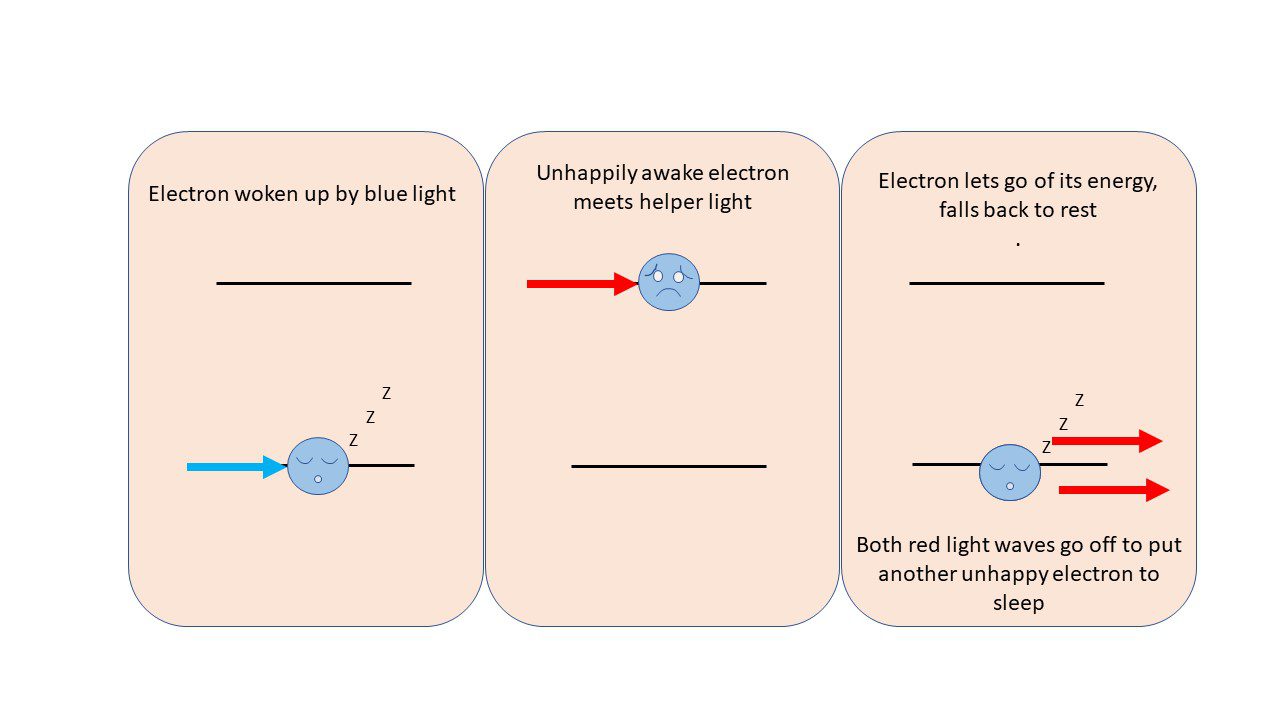
Dr. Maiman took advantage of this property and amplified it to make a very controlled beam of red light using this technique: If you shine blue light on a ruby, an electron in a chromium ion will absorb the light and the energy that comes with it, which bumps it up to a higher energy state. The electron would prefer to be as low energy as possible, and chromium electrons can reach this state by giving up a wave of red light.
But this isn’t all the red light that gets released. In fact, to eject the red light, the excited electron would prefer some help from a passerby waves of red light, cause this electron to vibrate at its own “red” frequency. With this encouragement, the electron will emit both the helper red light and some of its own energy as a beam of light with the a single, “red” frequency. Both of these red light waves can go on to stimulate the emission of more red light from other similarly excited electrons. In this way, the red light waves replicate over and over until you have a beam of light that is entirely… you guessed it, red – this is the laser light.
By leaving the original blue light on, you’ll keep powering up electrons to high energy states, and they keep converting that energy into red light as they calm down, helped along by all the other red light emission that’s going on around them.
This phenomenon, where light waves clone themselves over and over by helping excited electrons come down in energy, can happen in many different materials, not just ruby. And different materials emit different colors of laser light. In fact, ruby lasers are quite inefficient and aren’t used much today. (Sorry to say, the red light of the laser pointer you use to tease your cat is probably not from a ruby, but a diode. Diodes are a common components in computers, so scientists have figured out how to manufacture them so cheaply that they can produce millions at once – which is why laser pointers don’t cost anywhere near as much as a ruby necklace.)
From The Ruby Laser to Garnet Shields
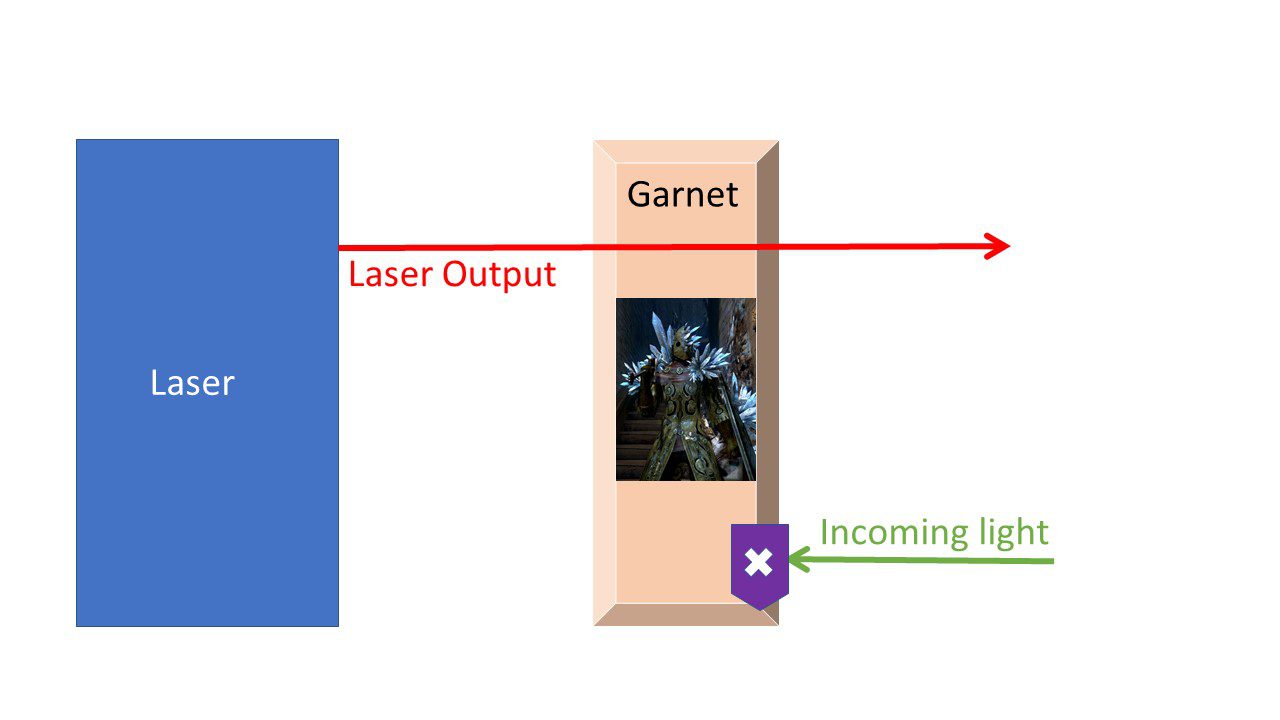
You may not think the laser “death ray” is a delicate system, but lasers, in fact, are quite finicky. As with all optical components, everything must be perfectly aligned, or else the light waves will not interfere constructively and they’ll cancel each other out with their randomness. In addition, scientists must blast lasers with a very specific amount of energy. And when a laser is producing its own light, this is a hard balance to strike: too much or too little light, and the entire laser is destabilized. For this reason, it’s useful to place a shield in front of the laser to protect it from light getting into the laser and causing problems.
This is where another popular gemstone comes in: garnets. These gemstones act like a tinted car window, but for laser light. They allow light to escape the laser, but they block light coming back towards it. This protects the delicate balance of the laser, just as a tinted car window protects your privacy. The one-way window lets light from outside in, allowing you to see the world, while blocking light leaving the car, so the world can’t see you. Garnets, therefore, are the courageous knights of optics, guarding the delicate, defenseless laser. What better way to honor their service than to wear some fine garnet jewelry?
Diamonds in electronics
In terms of wedding rings, diamonds are … well, they’re the diamond standard. You also may know them to be one of the hardest materials, and for this reason, scientists can use them to test the hardness of other materials. What you probably haven’t heard is how well diamonds conduct heat – five times better than copper, and better than any other naturally occurring material. This is useful in electronics, where small components handle lots of power and get very hot. To prevent melting or burning, computers require materials that move heat away from important electronic components as quickly as possible. While diamonds may be an expensive solution to this problem, they can be useful for research purposes, where scientists need the best materials available to support the most powerful devices.
Diamonds are composed of a carbon lattice held together by strong, unyielding bonds, which makes them highly resistant to changes in shape. In other words, diamonds are hard, so they can indent other materials without bending or breaking themselves. The strength of carbon bonds also contributes to the stiffness of diamond – the atoms are so rigidly held in formation that they would prefer to move together than to jiggle around individually. One application of this property is heat conduction: heat results from atoms vibrating vigorously, but in the case of diamond, like in a rigid, steel-framed car, any vibration will cause the whole network of atoms to vibrate.
A continuous vibration travelling through an entire network of atoms is called a phonon. Phonons transmit heat over large distances in diamond because diamonds are so pure, the long range vibrations aren’t interrupted and scattered into smaller, more local waves. Rather, they make it through the entire diamond, along with all of their heat energy.
Phonons can transmit the most energy in a lattice where the atoms are lightweight and the bonds are strong, and diamond excels at both of these properties. This type of heat conduction is useful in computer chips, where very small parts are all wedged together into a tiny space and can get quite hot. Engineers deposit synthetic diamonds under these computer components as heat spreaders to conduct the heat away, keeping the computer part from overheating. Next time you see a super computer, keep an eye out for some cool diamonds!
People have definitely recognized one of nature’s most special gifts when it comes to gemstones. They stand so far apart from other materials in their purity and the ways they interact with light, it’s no wonder they’re universally valued for their rarity and beauty. At the same time, exactly because they are so ordered and perfect, they’re great tools in physics research and engineering. So when you see some sparkling crystals, make sure to take a closer look! They may be just as appealing to your scientific side as they are to your inner fashionista.
Stella is a senior at Brown University studying materials engineering and researching polymer chemistry. She also loves knitting, dancing, and solving puzzles in her free time.