On April 8th, 2024, a total solar eclipse will sweep across North America, from Mexico to the Maine-Canadian border. For those who experienced the spectacular solar eclipse of 2017, this one will be similar, crossing the United States from west to east and passing...

Blood transfusions are an essential component of modern-day medicine, saving lives in a variety of situations, ranging from genetic diseases like sickle cell anemia to road accidents. But, the history of blood transfusion is a rocky one. For instance, did you know that a German physician founded the world’s first blood transfusion institute in 1926 because he believed blood transfusions led to immortality?
Dr. Alexander Bogdanov started some crude blood transfusion experiments on himself by injecting blood of other young men into his own system. After 11 such transfusion sessions, he claimed to have improved vision, arrest hair loss, and produce youthful skin. This led him to believe that blood transfusion was the path to immortality and eternal youth. But, as you can expect, this practice, combined with poor understanding of the blood transfusion process at that time, killed him years later.

The first major leap in our understanding of blood came from the discovery of blood circulation by William Harvey in the 17th century. He showed that the body was capable of processing both oxygen-rich and oxygen-lacking blood at the same time without mixing them up. In fact, through the coordinated action of the lungs and the heart, circulation ensures all of the body’s tissues are supplied with oxygen, and that carbon dioxide is consistently removed from the blood. Harvey’s discovery that blood has its own pathway through the body put to rest the theories behind many bizarre transfusion applications, like pouring the blood of one individual into the mouth of another.

Karl Landsteiner made the next pivotal discovery in our understanding of blood: the presence and nature of blood types. He described a blood type classification system based on the presence or absence of unique chemical molecules on red blood cells. This finding improved the success rates of human-to-human blood transfusion: physicians now understood why mismatches between blood types led to deaths.
The real ‘blood boom’ took place during World War I. Medical camps at war grounds needed a continuous supply of fresh blood to keep wounded soldiers alive. To meet the need, blood banking and storage gained prominence during this time. Blood banking became more efficient with the application of sodium citrate, a chemical with potent anti-clotting properties. Adding this chemical helped keep the blood fresh when refrigerated. This innovation made it possible to store, later transfuse blood from dead soldiers and helped save thousands of lives. In recent times, regular blood donation drives are organized by hospitals and NGOs to encourage everyone to do their part towards saving lives.
An average adult has 1 – 1.2 gallons of blood circulating in their body, a little more than the contents in a standard milk jug. Of this, in a single sitting, a person can donate enough to fill a can of soda. This amount of blood can save up to three lives. But while blood donation and transfusion are simple and safe for the most part, in some cases, the practice can cause further health problems. In one study, patients who received blood transfusions after heart surgery suffered from health complications, which led several organs to fail simultaneously. When researchers tried to understand the cause, they found that most of the patients whose condition deteriorated had received blood that had been in storage for a longer time.
They asked what made this blood so dangerous. They discovered that red blood cells contain a compound that transports nitric oxide gas, which helps blood to flow smoothly. When blood is stored for a long period of time outside the body, this compound degrades. After its transfusion, this blood sucks the nitric oxide out of the recipient’s body to make up for its low levels. This leads to a lack of oxygen in the body, and the tissues start to die, leading to stroke, cardiac arrest and organ failure in the recipient. Adding nitric oxide to stored blood circumvented this problem to a good extent. With improved storage conditions over the last decade, such complications have become rare.
To completely avoid transfusion-related complications, a potential solution that gained popularity was that of creating ‘artificial/synthetic’ blood. The use of blood substitutes such as milk can be dated back to the 1600’s. A popular option in the 1800’s was the use of Ringers solution: a solution of sodium, potassium, and calcium that, when mixed with lactate, restored people’s blood pressure. Ringers solution failed to sustain as a blood substitute because it could not carry oxygen as efficiently as red blood cells. Several alternatives were invented but all these systems were unable to provide everything blood could.
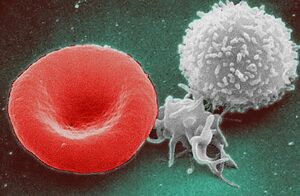
For example, blood protects the body from infection: white blood cells are on constant surveillance, looking out for incoming germs. Proteins in the blood identify injuries and form a mesh-like structure on the wound to prevent excessive bleeding. Synthetic blood lacks such intrinsically valuable blood components. Since most blood alternatives have failed so far, scientists are trying synthetic approaches, such as employing “nanomedicine.” Scientists are improving the composition of the blood alternatives by including in small particles (nanoparticles) containing or bound to important blood proteins that perform blood-like functions, such as protecting the body from infection. However, they haven’t achieved much success so far.
At an individual level, one way doctors can lower the risks associated with transfusion has been to store a surgery patient’s blood a few weeks prior to their surgery. Patients, however, have to be healthy enough to give large volumes of blood before their surgery, across multiple sittings. This is not a viable option for anyone who has a low count of red blood cells, for example.
With all our recent knowledge, one wouldn’t jump to extreme conclusions like Dr. Bogdanov did about transfusions causing immortality. Considering the current limitations on the efficiency of blood alternatives and transfusion, paired with every individual’s potential to donate blood, it is only fair we pledge a part of our heart and mind to do the same. Knowing how fragile life can be, the blood donor today might become a recipient tomorrow, making the gift of life come a full circle!